What is aldol condensation?
The aldol condensation, which is also referred to as the aldol reaction, is a chemical process that involves the acid- or base-catalyzed condensation of a carbonyl compound with the enolate or enol of another compound. These compounds may or may not be identical, and the reaction ultimately generates a β-hydroxy carbonyl compound, also known as an aldol.
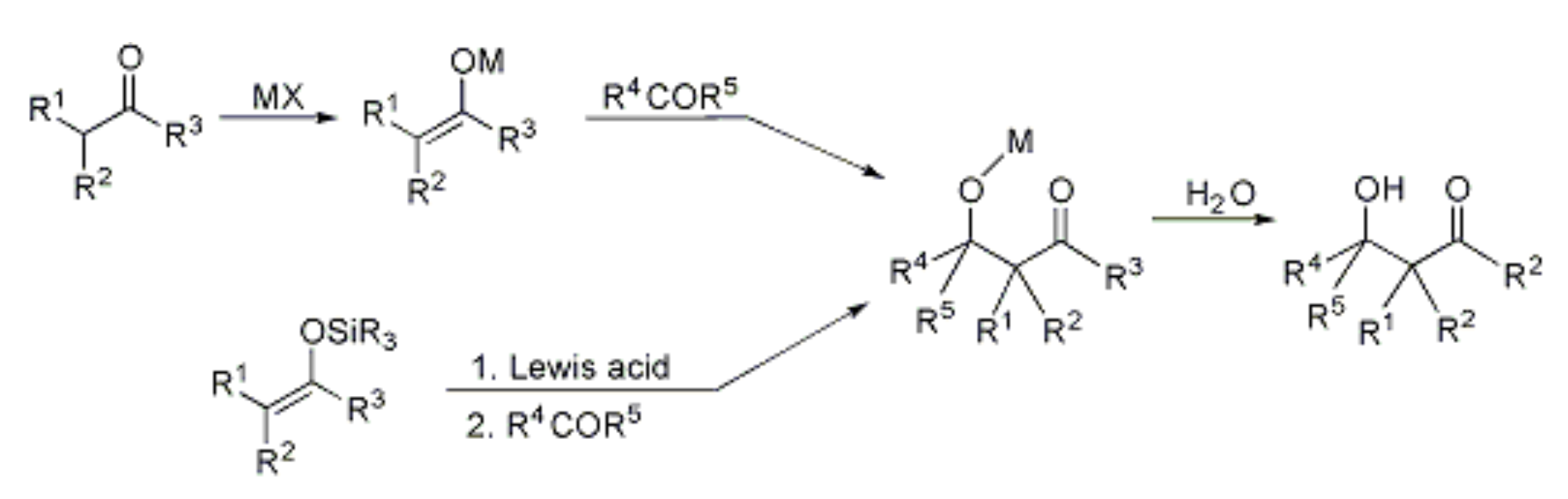
- MX = LDA, LHMDS, CH3MgBr, Bu2BOTf/NR3, 9-BBNCl/NR3, LDA + ZnCl2, LDA + (C5H5)2ZnCl2, LDA + T(O-i-Pr)3Cl. See list of acronyms.
- Lewis acid = TiCl4, SnCl4, AlCl3, BF3·OEt2, ZnCl2. See list of acronyms.
This method is hindered by several factors, including self-condensation, polycondensation, the formation of regioisomeric enols/enolates, and the dehydration of aldol followed by Michael addition. However, the development of techniques for preparing and utilizing preformed enolates or enol derivatives has significantly advanced the coupling of carbonyl compounds by allowing for the specific formation of carbon-carbon bonds.
References
- R. Kane, Ann. Phys. Chem., Ser. 2, 44, 475 (1838)
- Kane, R. (1838), Ueber den Essiggeist und einige davon abgeleitete Verbindungen. [On the vinegar spirit and some compounds derived from it] J. Prakt. Chem., 15: 129-155. https://doi.org/10.1002/prac.18380150112