Written by J.A Dobado | Last Updated on May 2, 2024
Objective
Extract photosynthetic pigments and separate them by a simple column chromatography (CC) and thin layer chromatography (TLC) techniques.
Background
Spinach leaves contain a variety of pigments that are important for the plant’s survival. These pigments play a crucial role in the process of photosynthesis. There are several different types of pigments (natural products) present in spinach leaves, including carotenoids, chlorophylls, xanthophylls, water-soluble vitamins, etc.
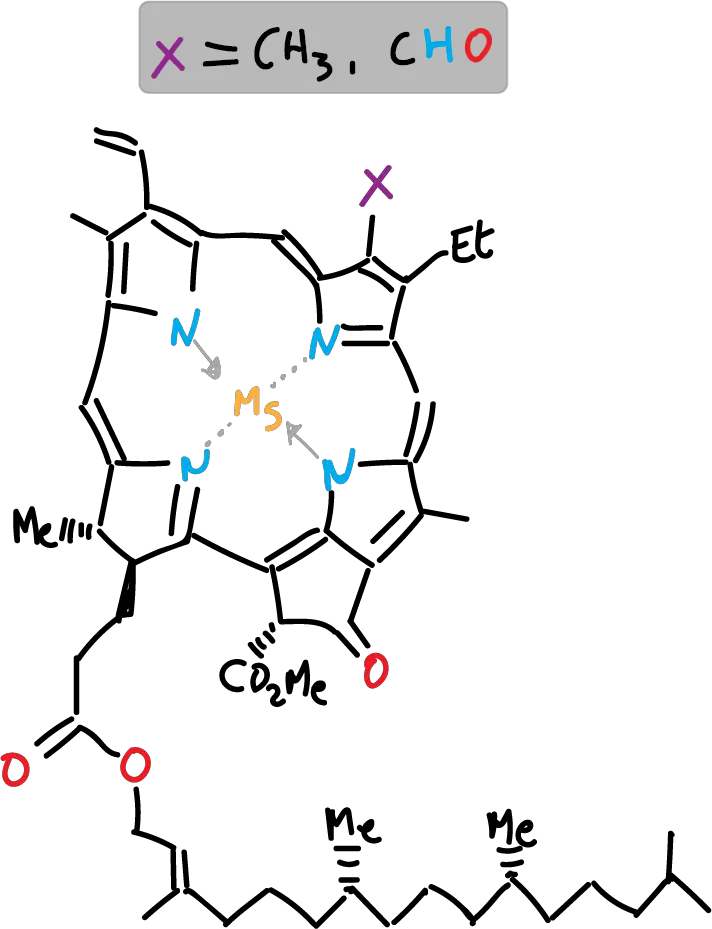
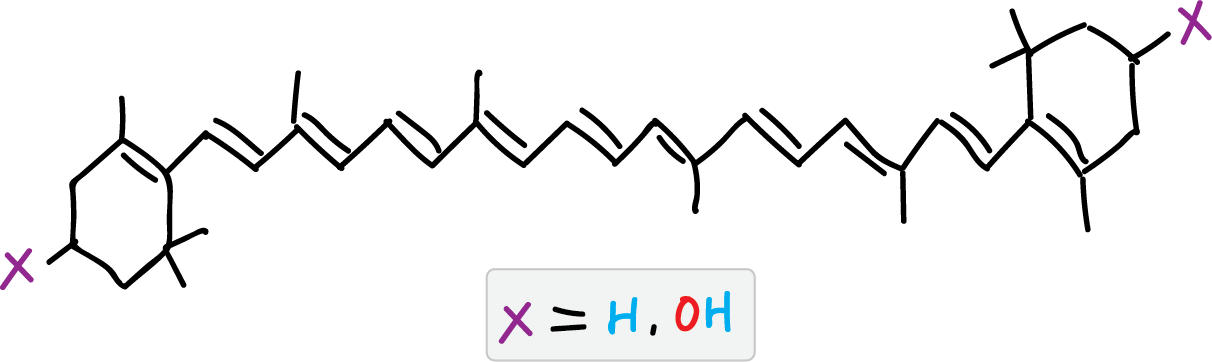
Spinach leaves have β-carotene and chlorophyll as the main components responsible for their color and smaller amounts of xanthophylls. The β-carotene has a yellow color, while chlorophyll has a green color, with two varieties, a and b, which differ in that a has a methyl group and b has a formyl group on the porphyrin ring. The loss of the Mg++ ion in chlorophylls leads to the formation of pheophytin (a and b). On the other hand, xanthophylls are a family of compounds, derived from carotenes with oxygen atoms in the structure and which in spinach leaf are found in smaller proportion than β-carotenes and chlorophyll.
In this experiment, pigments are extracted from spinach leaf and analyzed and separated by thin layer chromatography (TLC) and column chromatography (CC). Spinach chromatography allows the separation of substances from a mixture based on the different affinity that these products have for the stationary phase (silica gel) and a mobile phase (eluent). These techniques use a solvent to dissolve the pigments and then separating the resulting mixture based on their different solubilities and molecular structures.
Experimental procedure
A) Sample preparation
Weigh approx. 2 g of spinach leaves.[1] Wash the leaves with water, remove the nerves, chop them with scissors and put them in a mortar, together with 22 ml of acetone and 3 ml of hexane and add a small amount of calcium carbonate (CaCO3) with the help of a spatula (prevents the degradation of photosynthetic pigments). Grind the mixture until the leaves are discolored and the solvent acquires an intense green color.
Transfer only the obtained liquid to a separating funnel (avoid transferring solids), and add 20 ml of hexane and 20 ml of 10 % NaCl aqueous solution. Shake the mixture and let decant. The aqueous (lower) phase is discarded and the organic (upper) phase is washed with 5 ml of deionized water. It is decanted again, and the organic phase is transferred to a 50 ml Erlemeyer. The solution is dried over anhydrous sodium sulfate Na2SO4 and the desiccant is removed by filtration by gravity.
The sample is divided into two parts. 1 ml of the solution is taken and reserved in a test tube for the analysis of the sample by thin layer chromatography TLC and the rest is used for chromatographic separation on CC column.
B) Spinach chromatography by thin layer chromatography (TLC)
- Prepare a rectangle of filter paper with a height and perimeter lower than those of the TLC tank and place it against the wall of the inner part of the tank, making sure that throughout the experiment it allows a perfect view of the chromatogram.
- Prepare a 7:3 hexane:acetone solution.
- Introduce the eluent inside the TLC tank (only a volume that allows a solvent height inside the TLC tank of about 5 mm will be necessary) and cover the cuvette hermetically, to allow that, when impregnating the filter paper, the solvent vapors saturate the whole volume of the tank.
- With a pencil (do not use pens or markers, only graphite pencils) and a ruler draw a horizontal line about 5 mm from the base of a chromatofolio (Al TLC 5 x 7.5 cm silica gel 60F).[2]
- With the help of the glass capillary place a drop of the filtered liquid on the line drawn on the chromatoplate.
- Quickly insert the chromatofolio into the flask, in a vertical position, slightly inclined and in such a way that from the outside we have a view of the level of the solvent in the chromatofolio at all times.
- When the solvent level is within a few millimeters of the top of the chromatoplate, remove it and allow it to air dry. (Colored spots will appear along the plate).
- Mark with a pencil the level reached by the solvent.
- Quickly cover the TLC tank so that its atmosphere remains saturated with solvent.
- The appearance of colored spots along the plaque will be observed, so that carotenes will advance more rapidly and chlorophyll to a lesser degree.
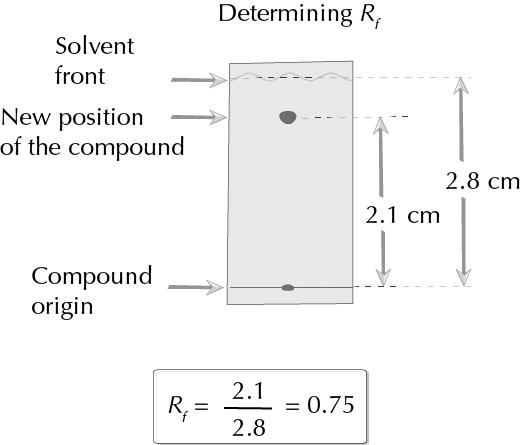
- Determine the value of Rf.
| Pigment | Color | Rf |
| Carotene | Yellow–Orange | 0.93 |
| Pheophytin a | Gray | 0.55 |
| Pheophytin b | Gray (may not be visible) | 0.47-0.54 |
| Chlorophyll a | Blue-Green | 0.46 |
| Chlorophyll b | Green | 0.42 |
| Xanthophylls | Yellow | 0.41, 0.31, 0.17 |
For the calculation of Rf see the following link.
C) Spinach chromatography by column chromatography (CC)
- Put some cotton or glass wool at the bottom of the column.
- Introduce 10 ml of eluent (hexane:acetone, 7:3) into the column.
- Weigh 20 g of silica gel 40 in a 50 ml Erlenmeyer flask and add a few ml of eluent, stirring with a glass rod or a spatula, until a slurry is formed.
- Fill the column with the slurry by opening the column tap, until the liquid is at the right limit of the stationary phase (it is very important that the column never remains dry, totally or partially, and that no air bubbles are formed), collecting the solvent in a container to be reused.
- Sand can be added to the silica gel to form a layer 1 or 2 centimeters thick over the boundary of the stationary phase.
- Take with a pipette, 10 ml of the sample prepared from the spinach leaves and add them to the column.
- Add eluent by opening the column stopcock and collecting the eluent in an Erlenmeyer flask.
- When a band of yellow coloration approaches (β-carotene) collect the liquid in a test tube.
- Change the test tube when the tube begins to fill or the contents of the tube change color.
Physico-chemical properties
This table collects data for the molecular weight (Mw), melting point (M.p.) boiling point (B.p.) and density of the reactives and compounds used in this laboratory experiment.
| Name | Mw (g/mol) | M.p. (ºC) | B.p. (ºC) | Density (g/ml) |
| Acetone | 58.08 | -94 | 56 | 0.791 |
| Hexane | 86.18 | -95 | 69 | 0.659 |
| CaCO3 | 100.09 | 800 | - | 2.93 |
| Chlorophyll a | 893.49 | 150-153 | - | - |
| Chlorophyll b | 907.47 | 183-185 | - | - |
| β-Carotene | 536.87 | 176-184 | - | 1.000 |
| NaCl | 58.44 | 801 | 1,413 | 2.165 |
| Na2SO4 | 142.04 | 884 | - | 2.630 |
GHS pictograms
Hazard pictograms form part of the international Globally Harmonized System of Classification and Labelling of Chemicals (GHS) and are collected in the followinf Table for the chemical compounds used in this experiment.
| Name | GHS |
| Acetone |   |
| Hexane |     |
| CaCO3 | Non-hazardous |
| Chlorophyll a | Non-hazardous |
| Chlorophyll b | Non-hazardous |
| β-Carotene | Non-hazardous |
| NaCl | Non-hazardous |
| Na2SO4 | Non-hazardous |
International Chemical Identifier
The IUPAC InChI key identifiers for the main compounds used in this experiment are provided to facilitate the nomenclature and formulation of chemical compounds and the search for information on the Internet for these compounds.
| Acetone | CSCPPACGZOOCGX-UHFFFAOYSA-N |
| Hexane | VLKZOEOYAKHREP-UHFFFAOYSA-N |
| CaCO3 | VTYYLEPIZMXCLO-UHFFFAOYSA-L |
| Chlorophyll a | ATNHDLDRLWWWCB-AENOIHSZSA-M |
| Chlorophyll b | NSMUHPMZFPKNMZ-VBYMZDBQSA-M |
| β-Carotene | OENHQHLEOONYIE-JLTXGRSLSA-N |
| NaCl | FAPWRFPIFSIZLT-UHFFFAOYSA-M |
| Na2SO4 | PMZURENOXWZQFD-UHFFFAOYSA-L |
References and notes
[1] This experiment can be performed with other green leaves if spinach is not available. It is also interesting to perform this experiment with other substances such as India ink. ↩
[2] The line should be above the solvent level when the chromatophore is introduced into the cuvette. ↩
[3] Isac-García, J.; Dobado, J. A.; Calvo-Flores, F. G.; and Martínez-García, H. (2015). Experimental Organic Chemistry Laboratory Manual. Elsevier Science & Technology. ISBN: 978-0-12-803893-2
[4] M. H. Anwar, Separation of plant pigments by thin layer chromatography, Journal of Chemical Education 40 (1963), no. 1, 29, DOI 10.1021/ed040p29.