Written by J.A Dobado | Last Updated on May 2, 2024
What is the Lewis structure of water H2O?
Lewis structures are diagrams that show how atoms in a molecule are arranged and bonded to each other. These diagrams are a helpful tool in chemistry to understand the bonding behavior of atoms and how they interact with each other. We will explain step-by-step how to draw the Lewis structure of water (H2O).
Step 1: Determine the total number of valence electrons
The first step in drawing the Lewis structure of water is to determine the total number of valence electrons present in the molecule. Valence electrons are the electrons in the outermost energy level of an atom that participate in chemical bonding. To determine the total number of valence electrons in water, we add up the valence electrons of each atom in the molecule.
Hydrogen (H) has 1 valence electron, and oxygen (O) has 6 valence electrons. Since there are two hydrogen atoms and one oxygen atom in water, the total number of valence electrons is:
2×(1) + 6 = 8
Step 2: Determine the central atom
The central atom is the atom that is bonded to the other atoms in the molecule. In the case of water, the oxygen atom is the central atom.
Step 3: Connect the atoms with single bonds
Next, we need to connect the atoms with single bonds. Hydrogen atoms can only form one bond, while oxygen atoms can form two bonds. Since water only has two hydrogen atoms, the oxygen atom will form one bond with each hydrogen atom. This gives us the following structure:
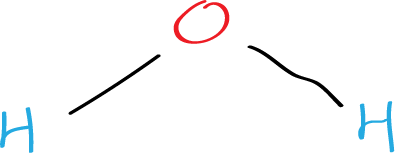
Step 4: Place the remaining electrons on the atoms
After connecting the atoms with single bonds, we need to place the remaining electrons on the atoms to satisfy the octet rule. The octet rule states that atoms tend to gain, lose, or share electrons to achieve a full outer shell of eight electrons (or two electrons for hydrogen). In the case of water, the oxygen atom needs two more electrons to achieve a full octet, while each hydrogen atom needs one more electron.
To satisfy the octet rule for the oxygen atom, we place two lone pairs of electrons around the oxygen atom. This gives the oxygen atom a total of eight valence electrons (4 electrons of the lone pairs and 4 electrons of the two single O-H bonds):

To satisfy the octet rule for each hydrogen atom, we do not need to add electrons to the hydrogen since it has two from the H-O bond.
Step 5: Check the formal charges
The formal charge is a measure of the distribution of electrons in a molecule. It is calculated by subtracting the number of non-bonding electrons and half of the bonding electrons from the number of valence electrons in an atom. The formal charge of an atom should be as close to zero as possible. To check the formal charges in water, we use the following formula:
Formal charge = Valence electrons – Non-bonding electrons – 1/2 (Bonding electrons)
For the oxygen atom, the formal charge is:
6 – 4 – 1/2(4) = 0
For each hydrogen atom, the formal charge is:
1 – 0 – 1/2(2) = 0
Since all the formal charges in water are zero, this confirms that our Lewis structure is correct.
Step 6: Draw the final structure
The final step is to draw the Lewis structure of water with all the lone pairs and bonds included:
