Written by J.A Dobado | Last Updated on April 22, 2024
What is the nomenclature of nitriles?
Nitriles are compounds that contain a carbon atom bonded to a nitrogen atom through a triple bond, which is represented by the formula R-C≡N (IUPAC recommendation C-831.1). The R group can be a variety of different functional groups, such as an alkyl group or an aryl group. Nitriles are important compounds in organic chemistry and are used in a wide range of chemical reactions and syntheses.
When the -C≡N group acts as a substituent, nitriles are usually named by adding the suffix “-nitrile” to the name of the parent compound. For example, the nitrile derived from propane is named “propionitrile.” When the parent compound is an aromatic compound, the suffix “-nitrile” is usually replaced with “-onitrile.” For example, the nitrile derived from benzene is named “benzonitrile.”
In simple cases they can be named in four ways (recommendation R-5.7.9):
- a) Add the nitrile termination to the name of the hydrocarbon with the same number of carbons.
- b) Consider it as a derivative of hydrocyanic acid (HCN), … cyanide.
- c) To name it as derivative of the acid of equal number of carbon atoms (including the -CN group).
- d) Consider it as acid derivative where -CN substitutes -COOH (carbonitrile). In this system, nitriles are named as derivatives of carboxylic acids, with the suffix “-carboxamide” being replaced with “-nitrile.” For example, the nitrile derived from propionic acid is named “propionitrile.”
The basic name of a nitrile is derived from the name of the corresponding carboxylic acid, by replacing the “ic acid” ending with “nitrile.” The name of the carboxylic acid is derived from the name of the corresponding hydrocarbon, with the suffix “-oic acid” added to the end.
For example, the nitrile of acetic acid is called acetonitrile, and the nitrile of propanoic acid is called propionitrile. The nitrile group is given the highest priority when numbering the carbon atoms, with the carbon of the nitrile group being assigned the number 1.
In addition to the basic names, nitriles can also be named using systematic names, which follow a set of specific rules. Systematic names for nitriles are typically derived from the name of the corresponding amide, by replacing the “-amide” ending with “-nitrile.”
For example, the nitrile of ethanamide is ethanenitrile, and the nitrile of propanamide is propanenitrile.
It is also important to note that nitriles can also be named using common names, which are often used in industry and in everyday conversation. Common names for nitriles are typically derived from the source of the compound or its properties.
Examples
There are also a number of common names for nitriles that are used in organic chemistry. For example, acetonitrile, which is derived from acetone, is commonly known as “methyl cyanide.” Similarly, benzonitrile is commonly known as “cyanobenzene” or “phenyl cyanide.”
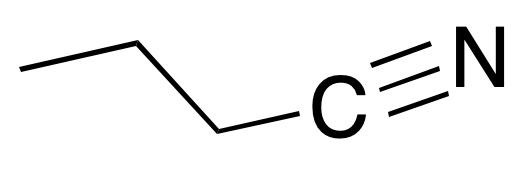
- Butanenitrile
- Propyl cyanide
- Butyronitrile
- Propanecarbonitrile
Other examples:
- Acetonitrile (basic name from acetic acid)
- Propionitrile (basic name from propanoic acid)
- Butyronitrile (basic name from butanoic acid)
- Valeronitrile (basic name from valeric acid)
- Ethanenitrile (systematic name from ethanamide)
- Propanenitrile (systematic name from propanamide)
- Butanenitrile (systematic name from butanamide)
- 2-Methylpropanenitrile (systematic name from 2-methylpropanamide)
- Phenyl cyanide (common name for benzonitrile)
- Ethyl cyanide (common name for ethylnitrile)
- Propyl cyanide (common name for propylnitrile)
- Isopropylnitrile (basic name from isopropanoic acid)
- Acrylonitrile (basic name from acrylic acid)
- Methacrylonitrile (basic name from methacrylic acid)
- Adiponitrile (basic name from adipic acid)
- Succinonitrile (basic name from succinic acid)
- Glutaronitrile (basic name from glutaric acid)
- Phthalonitrile (basic name from phthalic acid)
- Malononitrile (basic name from malonic acid)
Return to the page naming mono-functional compounds.