What is recrystallization in a Craig tube?
The Craig tube consists of two separable parts, an inner and an outer part. The outer part is similar to a test tube with rough ground glass in the widening tube neck. The plunger is usually made of Teflon or glass (in the latter case ground glass). Both parts fit together but allow liquids to pass between them even when closed.
This device is particularly suitable for recrystallizing amounts of solids ≤ 100 mg and solvent volumes ≤ 2 ml, minimizing transfers between containers, so solid loss in handling is also minimal.
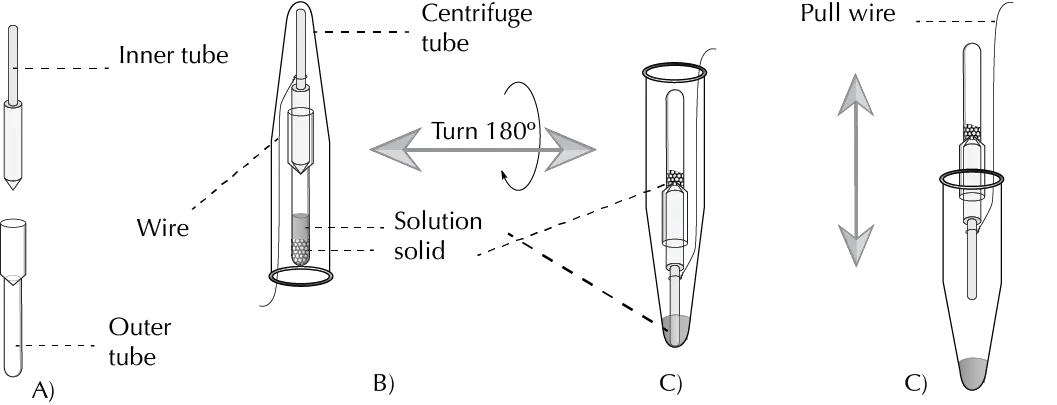
In the glass tube the solid to recrystallize is introduced, the solvent or mixture of solvents is added and it is heated in a water or sand bath until the crystals are hot dissolved. It is left to cool at room temperature until crystals appear.
Next, it is covered with the stopper. Then a small knot or loop is made with a piece of wire or thread of copper or other material inert to organic solvents, and it is covered with a centrifuge tube. The wire should be long enough to protrude from the centrifuge tube.
The centrifuge tube is then inverted so that the mother liquor from the recrystallization should fall to the bottom of the tube by sliding down the plunger and the crystals will be deposited in the widest part of the tube.
The glass tube is removed by hand and with the help of the wire the plunger is pulled gently to dispose of the crystals.
References
- Isac-García, J.; Dobado, J. A.; Calvo-Flores, F. G.; and Martínez-García, H. (2015). Experimental Organic Chemistry Laboratory Manual. Elsevier Science & Technology. ISBN: 978-0-12-803893-2
Back to the basic operations and specific microscale techniques page.