Written by J.A Dobado | Last Updated on April 22, 2024
Objective
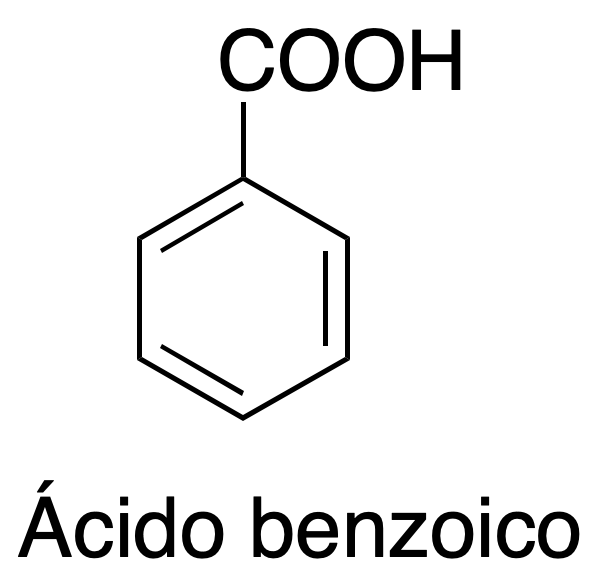
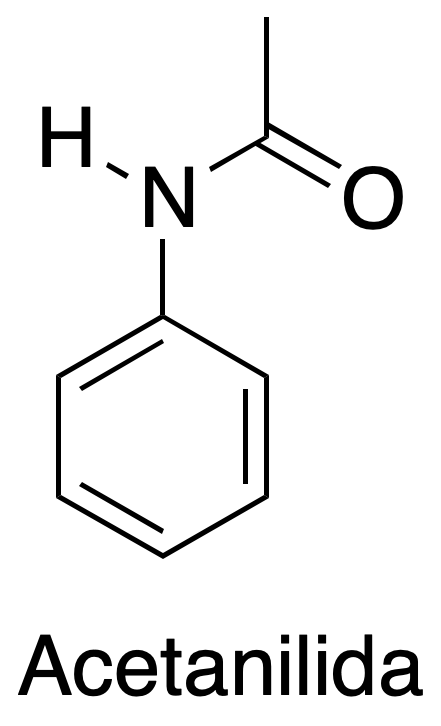
Study of recrystallization as a technique for purification and isolation of organic compounds in water and organic solvents, in this case we will use acetanilide and benzoic acid and as solvents water and hexane.
 |  |
 |  |
Background
To dissolve a solute in a solvent, the attractive solute-solvent interactions must be stronger than the intermolecular solute-solute and solvent-solvent attractions. The more similar these attractions are, the more easily attractive solute-solvent interactions will be established. In general, it can be said that “like dissolves like“.
- Nonpolar compounds dissolve in apolar solvents because the solute-solvent intermolecular forces of similar strength match those present in the undissolved solute (van der Waals interactions).
- Polar compounds dissolve in polar solvents because the solute-solvent intermolecular forces are of similar strength to those present in the undissolved solute.
- A nonpolar compound is not soluble in a polar solvent such as water, because nonpolar compounds can form hydrogen bonding interactions between the solute and solvent molecules.
Molecules that can form bonds (via hydrogen bonds) with water (hydrophilic) or that have ionizable groups (such as strong acids, acetic acid, MeOH, NaCl, etc.) will be soluble in water.
Experimental procedure
1) Recrystallization of acetanilide in water:
Weigh 1.5 g of acetanilide and transfer to a beaker. Add 35 ml of water and heat gently. Observe the appearance of an oil in the solution, which dissolves with the addition of several portions of hot water. If a colored solution is produced, add a small amount of activated charcoal (approximately 0.3 g), after heating and just before filtering. Filter the hot solution, quickly, with a conical funnel (using a pleated filter paper). Cool the mixture for about 30 minutes and filter out the crystals by vacuum filtration with the aid of the Büchner and Kitasato. Dry and weigh the resulting solid.
2) Recrystallization of benzoic acid in hexane:
Transfer 1.5 g of benzoic acid to a 250 ml round bottom flask and add 35 ml of hexane and a magnet. Connect a reflux condenser and heat the mixture to boiling with magnetic stirring. Keep refluxing until complete dissolution of the solid. Add activated carbon and filter while hot with a conical funnel (using a flutted filter). Collect the filtrate in a 100 ml Erlenmeyer flask and cover with a suitable stopper. Allow to cool at room temperature for 30 min and filter under vacuum with Büchner and Kitasato. Dry and weigh the resulting solid.
Physico-chemical properties
This table collects data for the molecular weight (Mw), melting point (M.p.) boiling point (B.p.) and density of the reactives and compounds used in this laboratory experiment.
| Name | Mw (g/mol) | M.p. (ºC) | B.p. (ºC) | Density (g/ml) |
| Acetanilide | 135.16 | 113-115 | 304 | - |
| Benzoic acid | 122.12 | 125 | 249 | 1.08 |
| Hexane | 86.18 | -95 | 69 | 0.659 |
GHS pictograms
Hazard pictograms form part of the international Globally Harmonized System of Classification and Labelling of Chemicals (GHS) and are collected in the followinf Table for the chemical compounds used in this experiment.
| Name | GHS |
| Acetanilide |  |
| Benzoic acid |   |
| Hexane |     |
International Chemical Identifier
The IUPAC InChI key identifiers for the main compounds used in this experiment are provided to facilitate the nomenclature and formulation of chemical compounds and the search for information on the Internet for these compounds.
| Acetanilide | FZERHIULMFGESH-UHFFFAOYSA-N |
| Benzoic acid | WPYMKLBDIGXBTP-UHFFFAOYSA-N |
| Hexane | VLKZOEOYAKHREP-UHFFFAOYSA-N |