Written by J.A Dobado | Last Updated on April 22, 2024
How are carboxylic acid anhydrides named according to IUPAC?
Carboxylic acid anhydride functional group is produced when two organic carboxylic acids combine and lose a water molecule. They are products of the dehydration of two carboxylic acids and the word anhydride means without water. When reacted again with water (hydrolysis reaction) they again form the starting carboxylic acids. The anhydrides have the grouping R-CO-O-CO-R’.
The most common ones are called symmetric when R = R’ and when such groups are different R ≠ R’ they are called mixed.
The IUPAC name is constructed from the source acid names (in alphabetical order) follow by the word anhydride (recommendations C-491.1 and C-491.2). The word acid is replaced by anhydride.
Examples
Symmetrical acid anhydrides
If we look at the first example in the figure, the central oxygen on both sides is the same group that would correspond to ethanoic (acetic) acid. Therefore, the name is constructed as ethanoic anhydride.
In the second example, the starting carboxylic acid would be propanoic acid, so the corresponding name for this molecule would be propionic anhydride.
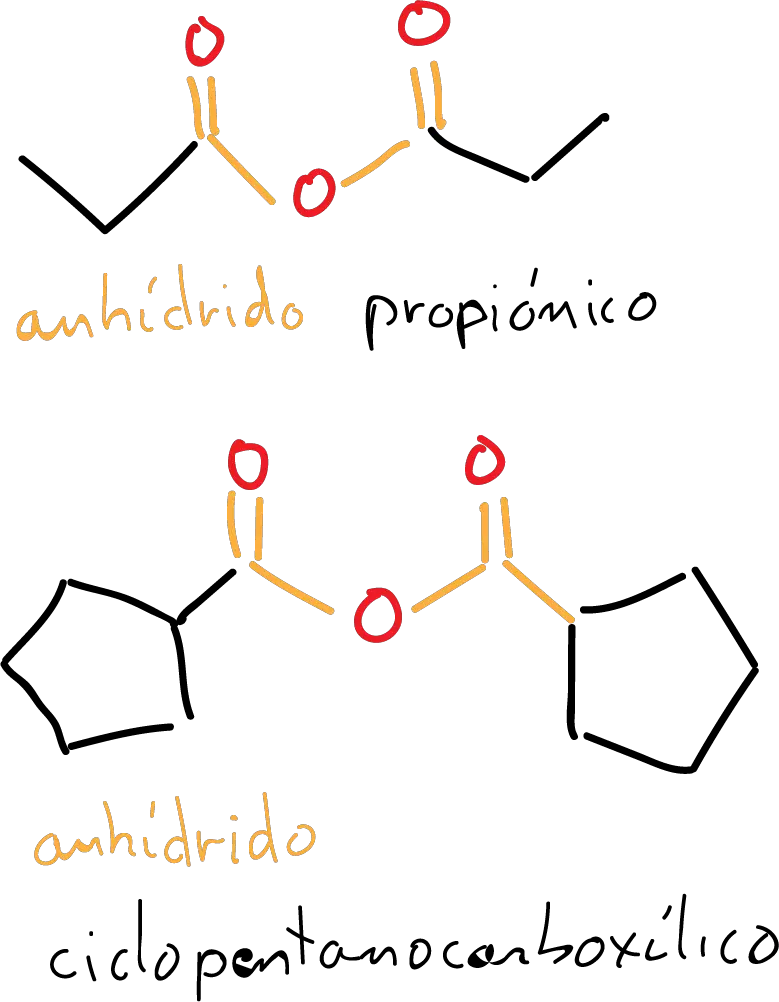
In the third example in the figure, the name is constructed from the name of the cyclopentanecarboxylic acid and would result in cyclopentanecarboxylic anhydride.
Asymmetric acid anhydrides
When the acid anhydride has different substituents, it is constructed by adding after the name that of the respective acids in alphabetical order eliminating the word acid.
In the first example in the figure, you start by naming by the acetic acid on the right of the structure and then continue with the formic acid on the left in the figure. That is, the resulting name would be acetic formic anhydride.
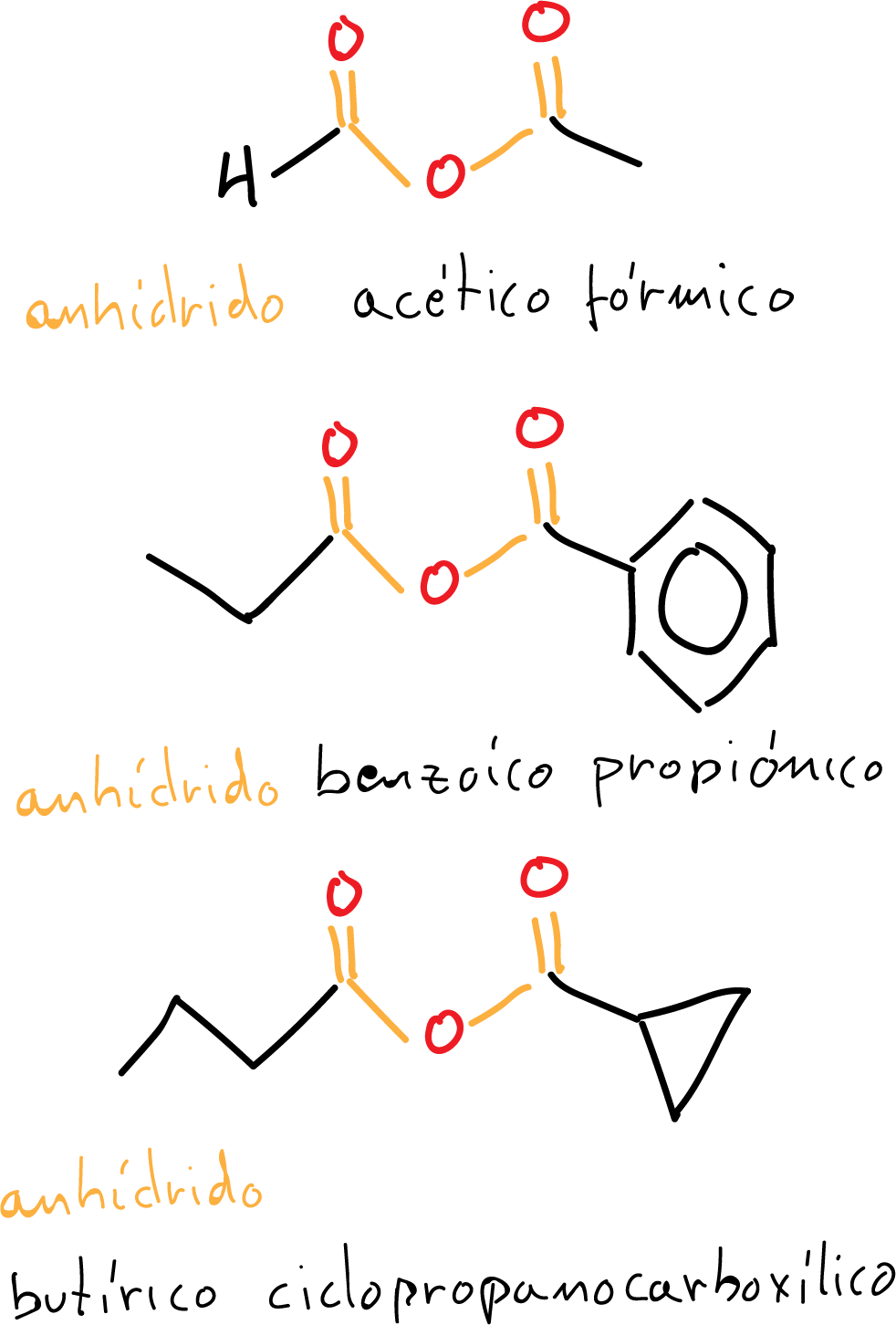
Following the same procedure, in the second example in alphabetical order it would be named as benzoyl propionic anhydride.
The molecule in the third example in the figure is named in the same way as the previous ones as butyric cyclopropanecarboxylic anhydride.
Trivial names of acid anhydrides
It is important to remember that many compounds in chemistry are still named using common names, in this case the common names of the carboxylic acids of origin prevail, so that the ethanoic anhydride is also named as acetic anhydride.
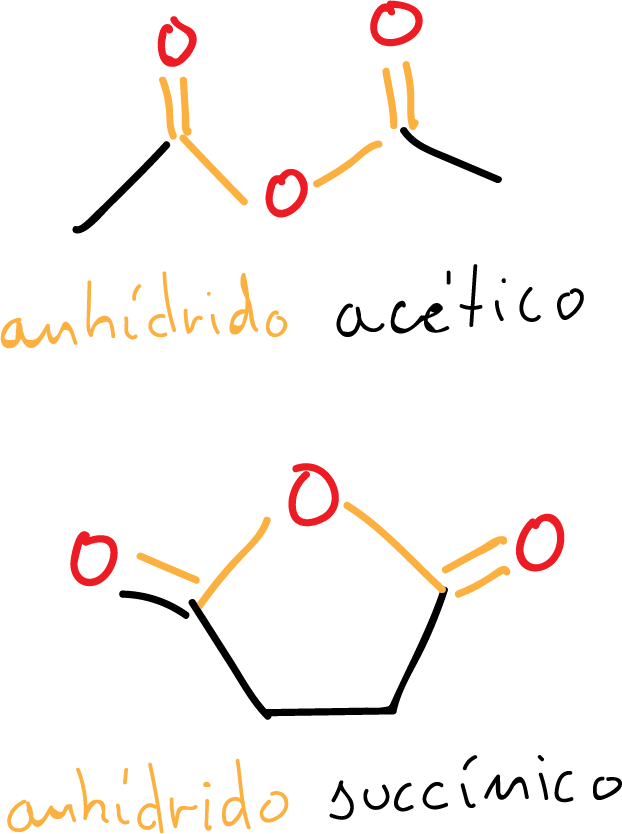
In the second example from the figure, the common name succinic anhydride is also used to denote butanedioic anhydride.
Return to the page naming mono-functional compounds.