Written by J.A Dobado | Last Updated on May 2, 2024
What is Blanc rule?
The Blanc rule was originally investigated by Lapworth in 1899 and later by Perkin in 1904 before being generalized by Blanc in 1907. Blanc reported that upon treatment with acetic anhydride, glutaric acid forms an intramolecular anhydride. Similarly, adipic and pimelic acids produce cyclopentanone and cyclohexanone respectively upon undergoing the same treatment. Based on these observations, Blanc concluded that when a dicarboxylic aliphatic acid is heated with acetic anhydride and distilled, the ketone of one less carbon atom would form, unless a five- or six-membered cyclic anhydride can be produced.
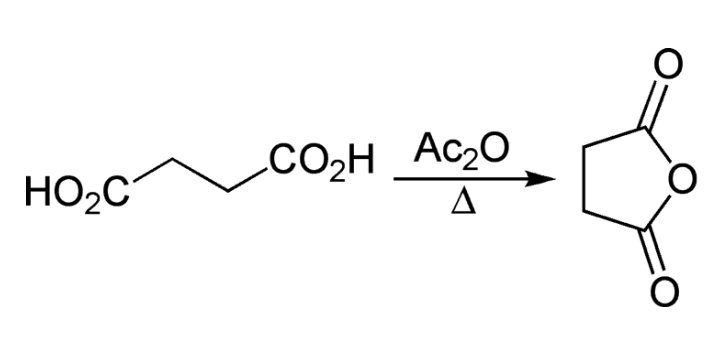
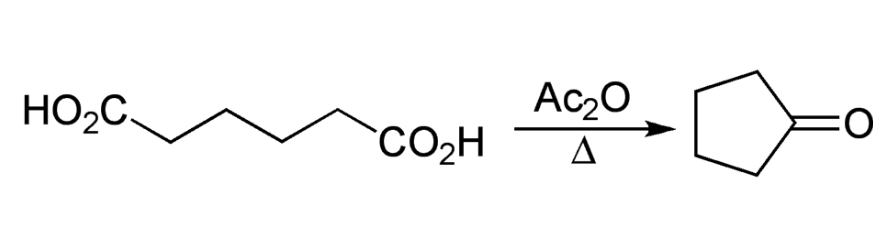
The cyclization reactions can be conducted without acetic anhydride by simply applying heat in a high vacuum. The Blanc rule has been widely applied in the constitutional analysis of the carbon skeleton of polycyclic systems, including sterols and bile acids, for a variety of substituted products of aliphatic dicarboxylic acids. However, it has been found to be unsuccessful in some cases, and an alternative method has been developed to differentiate cyclopentanones and cyclohexanones. This alternative rule involves heating dicarboxylic aliphatic acids to temperatures exceeding 200 ºC.
The Blanc rule has proven to be useful for characterizing five-membered or six-membered ketone rings in steroid skeletons, and it can also be utilized to prepare various cycloketones.
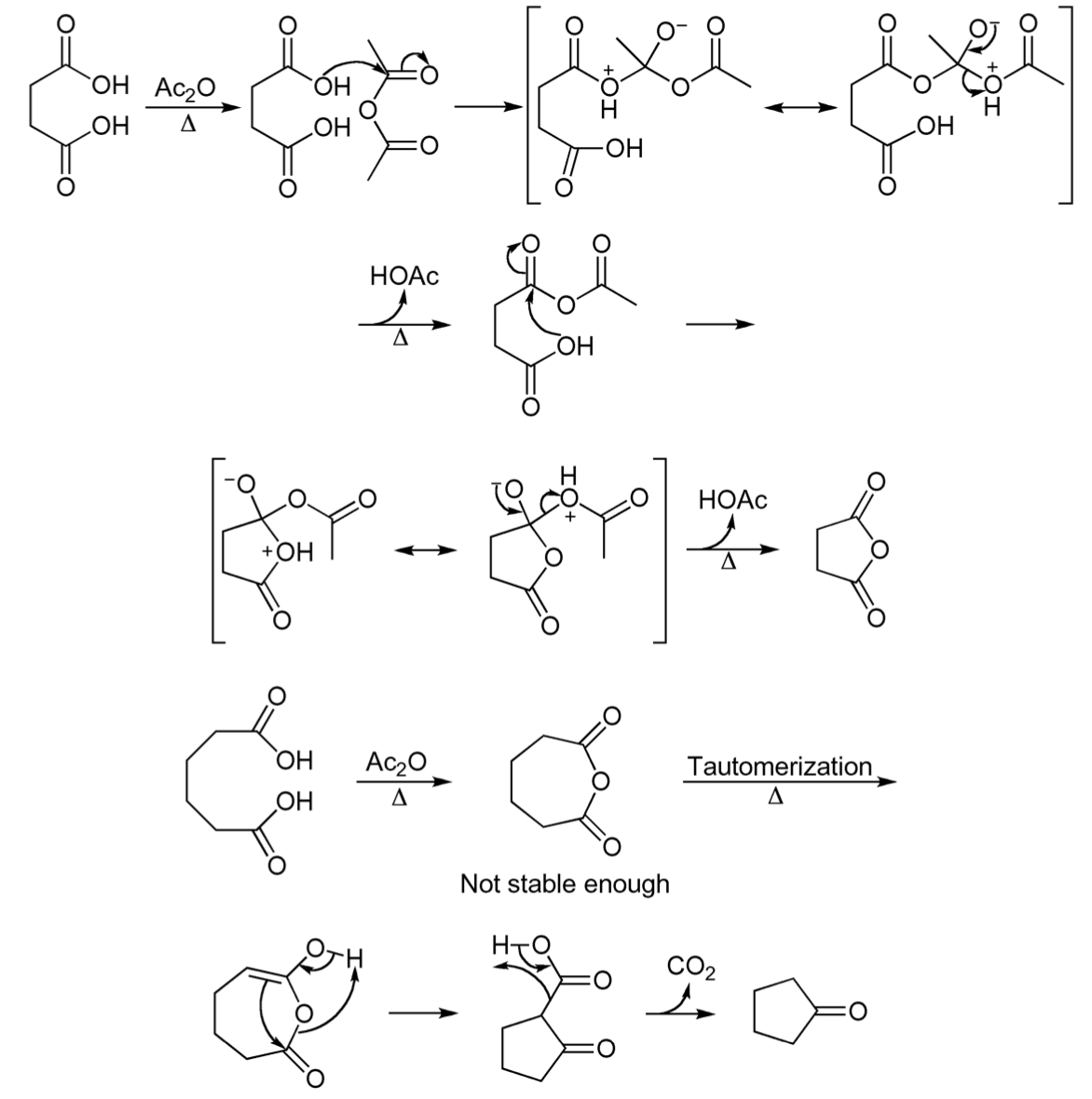
Mechanism of reaction
The mechanism depicted in the figure illustrates the process by which succinic anhydride and cyclopentanone are formed.
References
- Blanc, H.G., Compt. Rend., 1907, 144, 1356.
- Blanc, H.G., Bull. Soc. Chim., 1908, 3, 778.
- Lapworth, A.; Chapman, E.M., J. Chem. Soc., 1899, 1003.
- Lapworth, A.; Chapman, E.M., J. Chem. Soc., 1900, 464.
- Perkin, W.H.; Thorpe, J.F., J. Chem. Soc., 1904, 128.
- Perkin, W.H., J. Chem. Soc., 1904, 416.
- Perkin, W.H., J. Chem. Soc., 1906, 1640.