What is Gewald reaction?
In 1965, Gewald reported a reaction that involves a base-catalyzed condensation of a ketone with α-ketonitrile to produce an olefin that cyclizes with sulfur and is useful for synthesizing 2-aminothiophene. This reaction is known as the Gewald reaction and has various names such as Gewald synthesis of 2-aminothiophene, or Gewald aminothiophene synthesis.
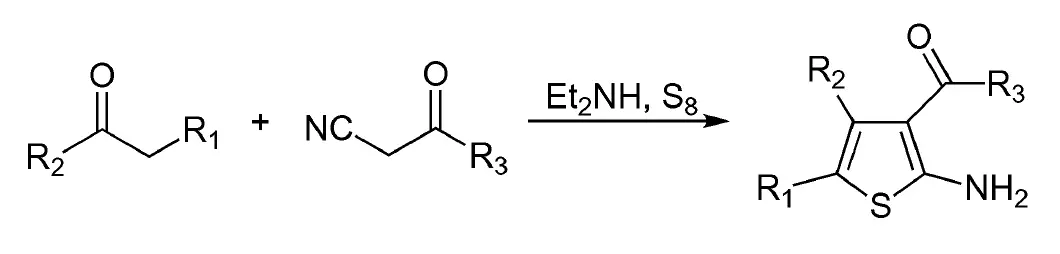
- R1 = alkyl, aryl
- R2 = alkyl, aryl
- R3 = OEt, OMe
The Gewald reaction has two major variants, a two-step synthesis and a one-pot synthesis, and diethyl amine is usually the catalyst. The reaction has been modified to synthesize 2-aminothiophene with a substituent at the 3-, 4-, or 5-position but is limited to the preparation of 2-aminothiophenes with electron-withdrawing groups at the 3-position and produces significant amounts of isomeric by-products if two active CH2 groups are available.
References
Gewald, K. (1965), Heterocyclen aus CH-aciden Nitrilen, VII. 2-Amino-thiophene aus α-Oxo-mercaptanen und methylenaktiven Nitrilen. [Heterocycles from CH-acid nitriles, VII. 2-amino-thiophenes from α-oxo-mercaptans and methylene-active nitriles.] Chem. Ber., 98: 3571-3577. https://doi.org/10.1002/cber.19650981120
Full Professor of Organic Chemistry at the University of Granada, with a long-standing research career in Computational Chemistry and molecular modeling and design.