Enolate formation
One of the main characteristics of the carbonyl group is that the hydrogens adjacent to the carbonyl carbon are acidic and can react with certain bases. If this occurs, the aldehyde or ketone becomes an enolate anion (nucleophile), which can react with a variety of electrophiles.
Furthermore, as the enolate ion is the result of two resonant structures, with the negative charge on oxygen or carbon, reactivity with electrophiles may be due to each of these, depending on the reaction conditions and the nature of the enolate and nucleophile (C- and O-alkylation).
fig1
Moreover, in an acidic medium, instead of forming the enolate ion, an enol is obtained, whose negatively charged carbon is susceptible to attack a protonated carbonyl.
fig2
Aldol condensation
One of the electrophiles that can react with the enolate anion is the carbonyl group of the reagents itself, since it has a positive partial charge. With this reaction a compound generically called aldol (β-hydroxycarbonyl) is obtained.
Aldol condensation consists of this reaction followed by an irreversible aldol dehydration step that generates an α,β-unsaturated carbonyl compound.
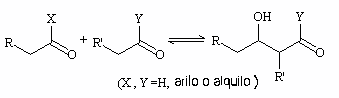
It can occur in acidic (via enol) or basic (via an enolate ion) media.
fig3
The dehydration step can occur in acidic or basic media, the difference is that in acidic media it takes place in a concerted manner in a single step (because H2O is a better leaving group than –OH).
fig4
In basic medium it proceeds in two stages (loss of the proton and elimination of HO⊖).
fig5
Crossed aldol condensation
The aldol condensation with acetone that we have seen gives only one (α,β-unsaturated) reaction product, because only one enolate was formed.
This can be complicated by reacting two different carbonyl compounds that each generate a different enolate and react, in turn, with the two reagents.
In this way four different aldols will be obtained, which, in turn can give different α,β-unsaturated carbonyl compounds with the subsequent dehydration step.
fig6
From the synthetic point of view, it is not very useful because product mixtures are produced.
Claisen-Schmidt condensation
The drawback of the mixture of products obtained in cross aldol reactions can be solved if this reaction is designed so that only one of the reactants forms the enolate anion and the other reactant is more electrophilic than the one that generates the enolate.
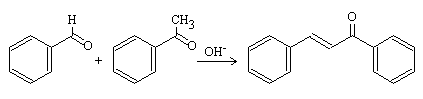
This is the case of Claisen-Schmidt condensation:[1]
fig7
Benzaldehyde is widely used for this type of reaction since it has no α-hydrogens and therefore cannot generate enolate ions.
In addition, the carbonyl group of benzaldehyde is much more electrophilic than that of acetaldehyde because it is attached to a charge-removing aromatic ring. Both requirements are met and a single α,β-unsaturated compound (cinnamaldehyde) is obtained.
Intramolecular aldol condensation
It is an aldol reaction that occurs between two carbonyl groups present in the same molecule to give a cyclic compound. 5- and 6-membered rings are favored over other ring sizes.
fig8
Claisen condensation
It is a condensation where the two reactants are esters, giving rise to a β-ketoester. Since the α-hydrogens of the ester are less acidic (pKa = 23-25) than those of an aldehyde or ketone (pKa = 19-20), a stronger base is needed to form the enolate (alkoxides).
fig9
The enolate ester reacts with another ester molecule to give a β-ketoester as indicated.
fig10
When the ester possesses only one α-hydrogen, no Claisen condensation product is observed. As in the case of crossed aldol condensation, Claisen condensation can take place between two different esters, being selective when one of them lacks α-hydrogens; ketones can also participate in Claisen condensation:
esquema1
Dieckmann condensation
It is an intramolecular Claisen condensation with which a β-ketoester is obtained, with the cyclic ketone and the ester group as the side chain of the ring.
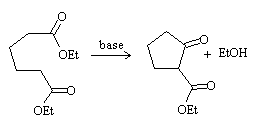
As in the case of intramolecular aldol condensation, the formation of 5- and 6-membered rings is favored.
fig11
Malonic ester and acetoacetic ester synthesis
Having considered the preparation of β-ketocarbonyl compounds, let us look at two examples of their synthetic utility. Since the α-hydrogens of β-ketocarbonyl compounds are more acidic (pKa = 9-13) than those of aldehydes, ketones and esters, a base such as alkoxides allows a proton to be captured from that position leading to the formation of an enolate in a quasi-quantitative manner. This enolate (which is nucleophilic) can be alkylated with a haloalkane to give substituted derivatives.
esquema2
Hydrolysis of a β-ketoester, gives 3-ketoacids which readily decarboxylate, under mild conditions, giving ketones. Hydrolysis of β-di diesters, likewise, also decarboxylates giving carboxylic acids:
esquema2
The combination of alkylation, hydrolysis and decarboxylation described above allows for the two strategies known as alkylation, hydrolysis and decarboxylation:
Acetoacetic ester synthesis (when starting from a β-ketoester) the sequence leads to substituted methylketones and malonic ester synthesis (when starting from a β-diester) the sequence produces substituted carboxylic acids.
esquema3
Michael addition
It is a 1,4- addition reaction where an enolate is added to conjugated carbonyl groups to give an α,β-unsaturated carbonyl compound.
fig12
Robinson annulation[2]
It is a particular case of Michael addition, followed by intramolecular aldol condensation, to give a cyclic compound.
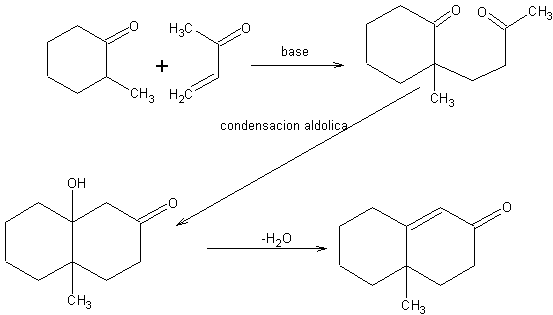
When a Michael addition occurs under very acidic or very basic conditions, it does not stop at the 1,5-dicarbonyl product, but continues to give an intramolecular aldol condensation between the two carbonyl groups.
fig13
Alkylation
The carbon of an alkyl halide is electrophilic in nature and can react with the enolate to generate alkylation products (C-C bond formation). These reactions have the disadvantage that neither HO⊖ nor RO⊖ can be used as bases, since they do not completely enolate the ketones and the mixture of products discussed above would be produced.
Moreover, being nucleophilic, they would react with alkyl halides. Consequently, bases such as LDA are used, which are strong (they completely convert ketones into enolates) and being very bulky, they are very little nucleophilic.
fig14
References and notes
[1] a) L. Claisen, A. Claparede, Chem. Ber. 14, 2460 (1881). b) J.G. Schmidt, Chem. Ber. 14, 1459 (1881)
[2] a) E.C. du Feu, F. J. McQuillin, R. Robinson, J. Chem. Soc. 1937, 53. b) A.L. Wilds, C. H. Shunk, J. Am. Chem. Soc. 65, 469 (1943).