What is Barton decarboxylation?
The Barton decarboxylation is a two-step process that involves the radical decarboxylation of organic acids to produce alkanes. In 1983, Barton first reported this reaction, which generates thiohydroxamic acid esters of the organic acid, followed by the addition of a radical initiator and reagents that provide a good H-atom donor, such as tri-n-butyltin hydride, t-butylmercaptan, phenylselenol, and tri(trimethylsilyl)silane. Barton decarboxylation is a chemical reaction that involves the removal of a carboxyl group (-COOH) from an organic compound.
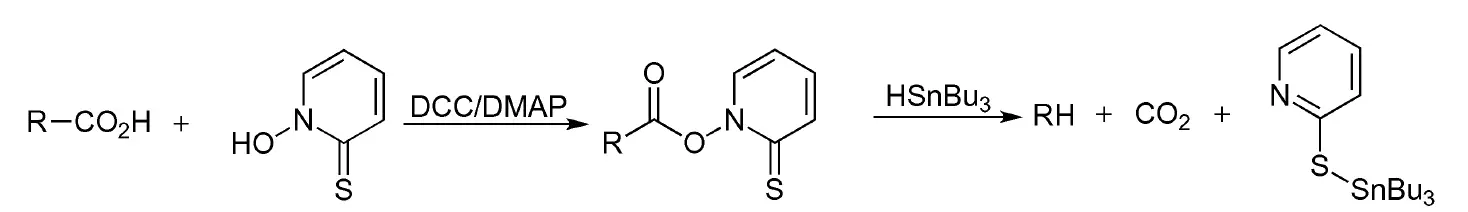
The Barton decarboxylation is also referred to as Barton’s radical decarboxylation. Notably, THF is not suitable as an H-atom donor, and the reaction can be initiated through photolysis or sonication, as well as through the use of a radical initiator (see list of acronyms).
References
- Barton, D. H. R., Crich, D., & Motherwell, W. B. (1983). New and improved methods for the radical decarboxylation of acids. J. Chem. Soc., Chem. Commun., (17), 939-941. https://doi.org/10.1039/C39830000939
- Barton, D. H. R.; Crich, D. and Motherwell, W. B., “Photolytic process for the formation of carbon-containing free radicals and its applications to free radical polymerization in particular” U.S. Patent, 1987, US 4668356
- Barton, D. H. R. and Zard, S. Z., “Invention of new reactions useful in the chemistry of natural products” Pure & Appl. Chem., 1986, 58, 675-684. DOI: 10.1351/pac198658050675
- Barton, D. H. R., Crich, D., and Motherwell, W. B. (1985). The invention of new radical chain reactions. Part VIII. Radical chemistry of thiohydroxamic esters; A new method for the generation of carbon radicals from carboxylic acids. Tetrahedron, 41(19), 3901-3924. https://doi.org/10.1016/S0040-4020(01)97173-X
Full Professor of Organic Chemistry at the University of Granada, with a long-standing research career in Computational Chemistry and molecular modeling and design.