Written by J.A Dobado | Last Updated on April 22, 2024
Objective
Isolation of caffeine from soluble coffee (from a commercial product), by extraction with an organic solvent and separation by centrifugation of the aqueous phase and the organic phase obtained.
Background
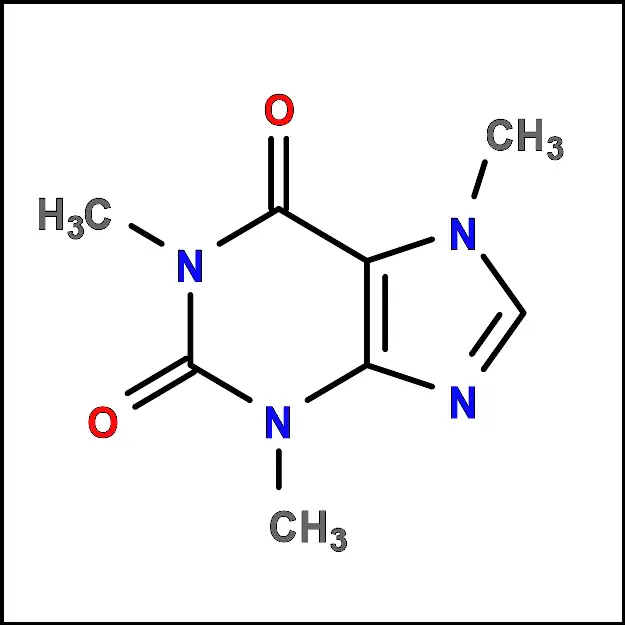
Caffeine is a naturally occurring alkaloid (xanthine family) found in a wide variety of plants, including coffee beans, tea leaves, cocoa beans, and kola nuts. When pure, it is an odorless white solid with a melting point of 234-236 ºC, partially soluble in water (100 mM). It is a stimulant that acts on the central nervous system, increasing alertness and reducing fatigue.
Instant coffee or soluble coffee is a product widely spread in the market that is obtained from infusion through the freeze-drying process.[1]
According to the manufacturers, instant coffee contains between 55 and 62 mg of caffeine for each cup prepared with a teaspoon of the product.
In this practice we will proceed to obtain caffeine from an extract obtained from a commercial product.
Once purified, caffeine can be used in a variety of applications, including the production of pharmaceuticals, beverages, and food additives. It is also used as a performance-enhancing supplement in sports and as a psychoactive recreational drug.
Overall, the isolation of caffeine is a crucial process that allows for the production and use of this widely consumed compound.
Experimental procedure
The isolation of caffeine from soluble coffee is carried out as follows:
In a beaker place 2 g of sodium carbonate and 2 g of instant soluble coffee.[2] Add 9 ml of very hot deionized water and stir the mixture until boiling. The mixture is then allowed to cool to room temperature.
The content of the beaker is poured into a 15 ml centrifuge tube, 2 ml of methylene chloride (CH2Cl2) is added, the tube is closed and shaken vigorously for 1 min. It is then centrifuged for 90 s. The yellowish CH2Cl2 solution is separated from the aqueous phase with the aid of a Pasteur pipette. The operation is repeated two more times.[3]
The organic extracts are pooled and dried over anhydrous sodium sulfate (Na2SO4). The solid is eliminated by filtration by gravity. The desiccant in the paper filter is washed with a few ml more solvent and pooled with the remaining CH2Cl2. The solution is transferred to a previously tared flask, the solvent is removed at the rotary evaporator and finally the flask is weighed and the caffeine obtained is calculated. The caffeine can be purified by EtOH recrystallization.
Physico-chemical properties
This table collects data for the molecular weight (Mw), melting point (M.p.) boiling point (B.p.) and density of the reactives and compounds used in this laboratory experiment.
| Name | Mw (g/mol) | M.p. (ºC) | B.p. (ºC) | Density (g/ml) |
| Caffeine | 194.19 | 234-236.5 | - | 1.230 |
| CH2Cl2 | 84.93 | -97 | 40.0 | 1.33 |
| EtOH | 46.07 | -114.1 | 78.5 | 0.790 |
| Na2CO3 | 105.99 | 851 | - | 2.532 |
| Na2SO4 | 142.04 | 884 | - | 2.630 |
GHS pictograms
Hazard pictograms form part of the international Globally Harmonized System of Classification and Labelling of Chemicals (GHS) and are collected in the followinf Table for the chemical compounds used in this experiment.
| Name | GHS |
| Caffeine |  |
| CH2Cl2 |  |
| EtOH |  |
| Na2CO3 |  |
| Na2SO4 | Non-hazardous |
International Chemical Identifier
The IUPAC InChI key identifiers for the main compounds used in this experiment are provided to facilitate the nomenclature and formulation of chemical compounds and the search for information on the Internet for these compounds.
| Caffeine | RYYVLZVUVIJVGH-UHFFFAOYSA-N |
| CH2Cl2 | YMWUJEATGCHHMB-UHFFFAOYSA-N |
| EtOH | LFQSCWFLJHTTHZ-UHFFFAOYSA-N |
| Na2CO3 | CDBYLPFSWZWCQE-UHFFFAOYSA-L |
| Na2SO4 | PMZURENOXWZQFD-UHFFFAOYSA-L |
References and notes
- [1] Process in which the product is frozen and then vacuumed to eliminate water by sublimation.
- [2] In this way we ensure that none of the hydrogens will be protonated, which will facilitate their dissolution in an organic solvent.
- [3] Alternatively, separation of the two phases by decantation can be performed, but is less effective.
- Isac-García, J.; Dobado, J. A.; Calvo-Flores, F. G.; and Martínez-García, H. (2015). Experimental Organic Chemistry Laboratory Manual. Elsevier Science & Technology. ISBN: 978-0-12-803893-2
- D. L. Pavia, Coffee, tea, or cocoa. A trio of experiments including the isolation of theobromine from cocoa, Journal of Chemical Education 50 (1973), no. 11, 791, DOI: 10.1021/ed050p791