What is Chan-Evans-Lam cross coupling?
The Chan-Evans-Lam cross coupling, also kown as Chan-Lam coupling, enables the formation of aryl carbon-heteroatom bonds by using oxidative coupling of arylboronic acids, stannanes, or siloxanes with compounds that contain N-H or O-H in the presence of air.
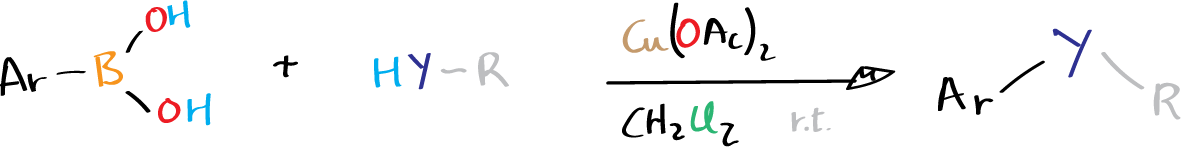
Y = NR’, O, S, NCOR’, NSO2R’
The substrates that can be used for this reaction include phenols, amines, anilines, amides, imides, ureas, carbamates, and sulfonamides. Copper(II) in a stoichiometric amount or a catalytic amount of copper catalyst can induce the reaction and get reoxidized by atmospheric oxygen. One advantage of the Chan-Evans-Lam cross coupling is that it can be carried out at room temperature in air, which sets it apart from the Buchwald-Hartwig cross-coupling.
Oxygen also aids in the reaction that involves a stoichiometric amount of copper(II) since the reductive elimination from a copper(III) species is more rapid.
References
- Chan, D.; Monaco, K.; Wang, R.; Winter, M. (1998). “New N- and O-Arylations with Phenylboronic acids and Cupric Acetate“. Tetrahedron Lett. 39 (19): 2933-2936
DOI: 10.1016/s0040-4039(98)00503-6 - Evans, D.; Katz, J.; West, T. (1998). “Synthesis of Diaryl Ethers through the Copper-Promoted Arylation of Phenols with Arylboronic Acids. An Expedient Synthesis of Thyroxine“. Tetrahedron Lett. 39 (19): 2937-2940
DOI: 10.1016/s0040-4039(98)00502-4 - Lam, P.; Clark, C.; Saubern, S.; Adams, J.; Winters, M.; Chan, D.; Combs, A. (1998). “New Aryl/Heteroaryl C-N Bond Cross-coupling Reactions via Arylboronic Acid/Cupric Acetate Arylation“. Tetrahedron Lett. 39: 2941-2944
DOI: 10.1016/s0040-4039(98)00504-8 - Lam, P.; Bonne, D.; Vincent, G.; Clark, C. (2003). “Copper-promoted/catalyzed C-N and C-O Bond Cross-coupling with Vinylboronic Acid and Its Utilities“. Tetrahedron Lett. 44: 4927-4931
DOI: 10.1016/s0040-4039(03)01037-2 - Chan, D.; Monaco, K.; Li, R.; Bonne, D.; Clark, C.; Lam, P. (2003). “Copper Promoted C-N and C-O Bond Cross-coupling with Phenyl and Pyridylboronates“. Tetrahedron Lett. 44: 3863-3865
DOI: 10.1016/s0040-4039(03)00739-1