What is Cannizzaro reaction?
The Cannizzaro reaction is a chemical reaction that involves the disproportionation of an aldehyde, in which an aldehyde is converted into an alcohol and a carboxylic acid. This reaction was first reported in 1853 and is named after the Italian chemist Stanislao Cannizzaro, who first described the reaction. It involves the use of concentrated NaOH or other strong bases, which result in the formation of an equimolar mixture of a primary alcohol and a carboxylic salt. However, this reaction is limited to aldehydes without α-hydrogens, as those with α-hydrogens undergo aldol condensation instead. As a result, the Cannizzaro reaction is commonly referred to as the Cannizzaro disproportionation.
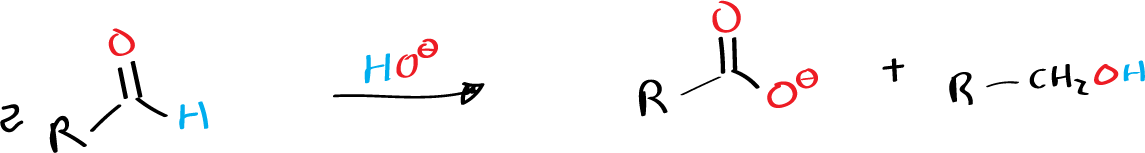
The hydride transfer mechanism in this reaction occurs directly between aldehydes, without the involvement of a reaction medium. Interestingly, the reaction can be utilized in the synthesis of benzyl benzoate from benzaldehyde. When the oxidant aldehyde differs from the reductant aldehyde, the reaction is known as the cross (or crossed) Cannizzaro reaction. This type of reaction is useful for synthesizing high yields of alcohol, such as when formaldehyde is reacted with another aldehyde in the presence of Ba(OH)2·8H2O at 100-110 ºC, resulting in more than 80-90 % alcohol yield without the need for a solvent.
In a crossed Cannizzaro reaction, aldehydes with electron-withdrawing substituents undergo the reaction at a faster rate than those with electron-donating groups. Different aromatic aldehydes can also be used, with the electron-donating aldehyde being reduced, and the electron-withdrawing aldehyde being oxidized. The susceptibility of various studied aldehydes to oxidation decreases in the following order: m-nitrobenzaldehyde, furfural, p-bromobenzaldehyde, benzaldehyde, and p-anisaldehyde.
The Cannizzaro reaction can occur under high-temperature and high-pressure conditions, such as 250 ºC and 4 MPa, to produce more than 50 % methanol MeOH from formaldehyde CH2O, even without the use of a catalyst. It can also occur on a solid surface, such as MgO and TiO2, and in the gas phase.
The Cannizzaro reaction is typically performed in the presence of a strong base, such as sodium hydroxide NaOH or potassium hydroxide KOH. When an aldehyde is treated with a strong base, it is converted into its conjugate base, which is a carbanion. This carbanion can then react with a molecule of the aldehyde, forming an alkoxide ion. The alkoxide ion is then converted into an alcohol by the loss of a proton, while the remaining molecule of the aldehyde is converted into a carboxylic acid through the loss of a proton and the addition of a hydroxyl group..
One of the key features of the Cannizzaro reaction is that it is an example of a self-sustaining process, meaning that the products of the reaction can participate in the reaction to produce more products. This leads to a runaway reaction, in which the aldehyde is rapidly consumed and converted into the final products..
The Cannizzaro reaction is often used in the synthesis of alcohols and carboxylic acids, and it can be used to synthesize a wide range of compounds with different functional groups. However, it has largely been replaced by more efficient and selective methods, such as the Baeyer-Villiger oxidation, which allows for the selective conversion of an aldehyde into a ketone or ester..
Summary
The Cannizzaro reaction is a useful method for the synthesis of alcohols and carboxylic acids, but it has largely been superseded by more modern methods. Despite this, it remains an important part of the history of chemistry and continues to be studied by chemists today..
Example
Here is an example of the Cannizzaro reaction:
2-Pentanone (an aldehyde) is treated with sodium hydroxide (a strong base)..
2-Pentanone + NaOH –> 2-Pentanol + Sodium formate
In this example, the aldehyde 2-pentanone is reduced to the alcohol 2-pentanol via the Cannizzaro reaction. The sodium formate is a byproduct of the reaction..
See an example of organic chemistry experiment with benzoic acid as the starting material in a Cannizzaro reaction.
Mechanism of reaction
The mechanism of the Cannizzaro reaction involves several steps:
- Protonation of the aldehyde: The aldehyde is protonated by the strong base, forming an aldehyde cation..
- Deprotonation of the aldehyde cation: The aldehyde cation is deprotonated by another molecule of the strong base, forming an alkoxide anion..
- Reductive deprotonation: The alkoxide anion is reduced by a protonated molecule of the aldehyde, forming an alcohol and a protonated molecule of the strong base..
- Deprotonation of the protonated strong base: The protonated molecule of the strong base is deprotonated, forming the final product (the alcohol) and a molecule of the strong base..
Overall, the Cannizzaro reaction involves the reduction of an aldehyde to an alcohol by a strong base, with the formation of a byproduct (such as sodium formate in the case of sodium hydroxide). The reaction requires two molecules of the strong base for each molecule of aldehyde..
References
- Cannizzaro, S. (1853), Ueber den der Benzoësäure entsprechenden Alkohol. [On the alcohol corresponding to benzoic acid.] Justus Liebigs Ann. Chem., 88: 129-130. https://doi.org/10.1002/jlac.18530880114