What is Emmert reaction?
The Emmert reaction, first reported by Emmert and Asendorf in 1939, involves the condensation of pyridine (or its analogues) with aldehydes or ketones in the presence of aluminium or magnesium amalgam to form pyridylalkyl or pyridyldialkyl carbinols. Pyridine and its analogues are typically considered unreactive towards alkylation or acylation reactions due to the presence of the C=N bond in the pyridine ring.
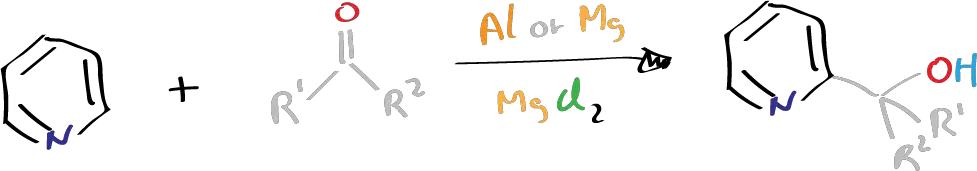
Despite pyridine being considered unreactive towards alkylation, it has been discovered that treatment of aldehydes or ketones with aluminium or magnesium, along with mercuric chloride HgCl2 and iodine, or with small amounts of mercuric chloride and a few drops of mercury, can lead to pyridine alkylation. Other metals with low reduction potentials do not work in this reaction, and while metals with high reduction potentials and poor coordinating power do not offer the alkylation product, they do offer the pinacols of ketones.
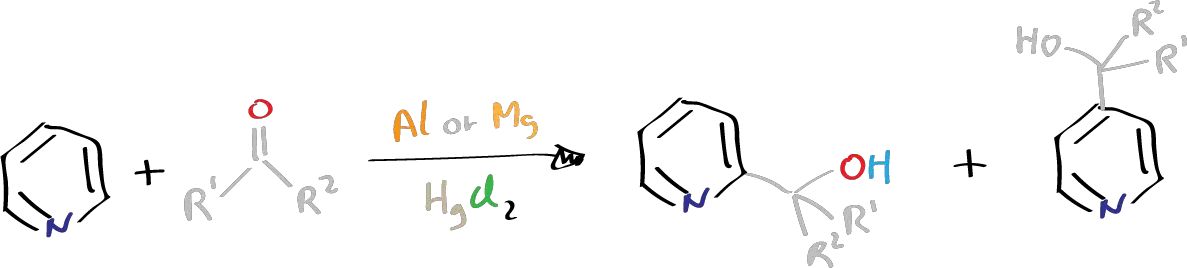
Magnesium or aluminium, which have high reduction potentials (1.7 V and 2.4 V, respectively) and coordinating power, are effective for this reaction. Beryllium (1.69 V) has similar properties but yields very poor results. Pyridine can react with dialkyl, diaryl, alkyl aryl, and cyclic ketones in this reaction, but steric effects from ortho substituents on the pyridine can inhibit the reaction between such pyridine and aryl ketones.
The precise role of the metal in this reaction is not entirely clear, but it is believed that the metal provides two electrons to the carbonyl compound, resulting in a carbanion that attacks the pyridine ring. Thus, an electron-donating group on the ketone can increase the negativity of a carbanion, making it easier to add to the pyridine ring. Additionally, ketones tend to give better yields than aldehydes due to the hyperconjugation of the alkyl group on the ketone.
Mechanism of reaction
The mechanism of the Emmert reaction begins with the donation of two electrons bye the metal to form the carbanion, which adds to pyridine ring, as depicted in the figure by the reaction of Mg.
References
- Emmert, B. and Asendorf, E. (1939), Eine Synthese von α-Pyridyl-dialkyl-carbinolen. [A synthesis of α-pyridyl-dialkyl-carbinols.] Ber. dtsch. Chem. Ges. A/B, 72: 1188-1194. https://doi.org/10.1002/cber.19390720610
- Emmert, B. and Pirot, E. (1941), Eine Synthese von α-Pyridyl und α-Chinolyl-dialkyl-carbinolen. [A synthesis of α-pyridyl and α-quinolyl-dialkyl-carbinols.] Ber. dtsch. Chem. Ges. A/B, 74: 714-719. https://doi.org/10.1002/cber.19410740510
Full Professor of Organic Chemistry at the University of Granada, with a long-standing research career in Computational Chemistry and molecular modeling and design.