What are alcohols?
Alcohols are compounds presenting the -OH functional group.
Acid-base properties
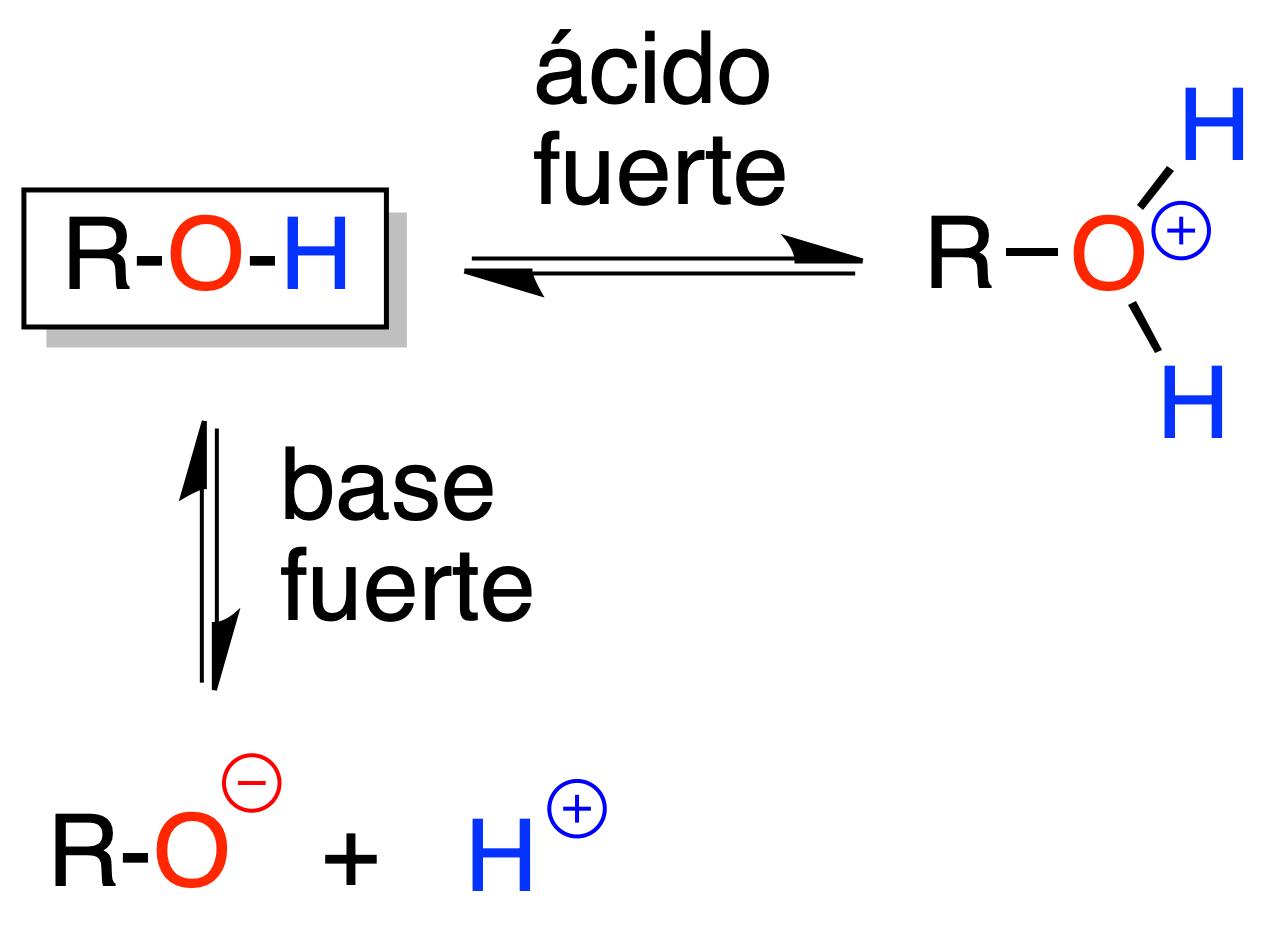
Alcohols can behave as acids or bases. Many reactions in which they are involved are justified according to their acid-base properties.
With strong acids they yield alkoxonium ions and with strong bases, alkoxides. Both are equilibrium processes.
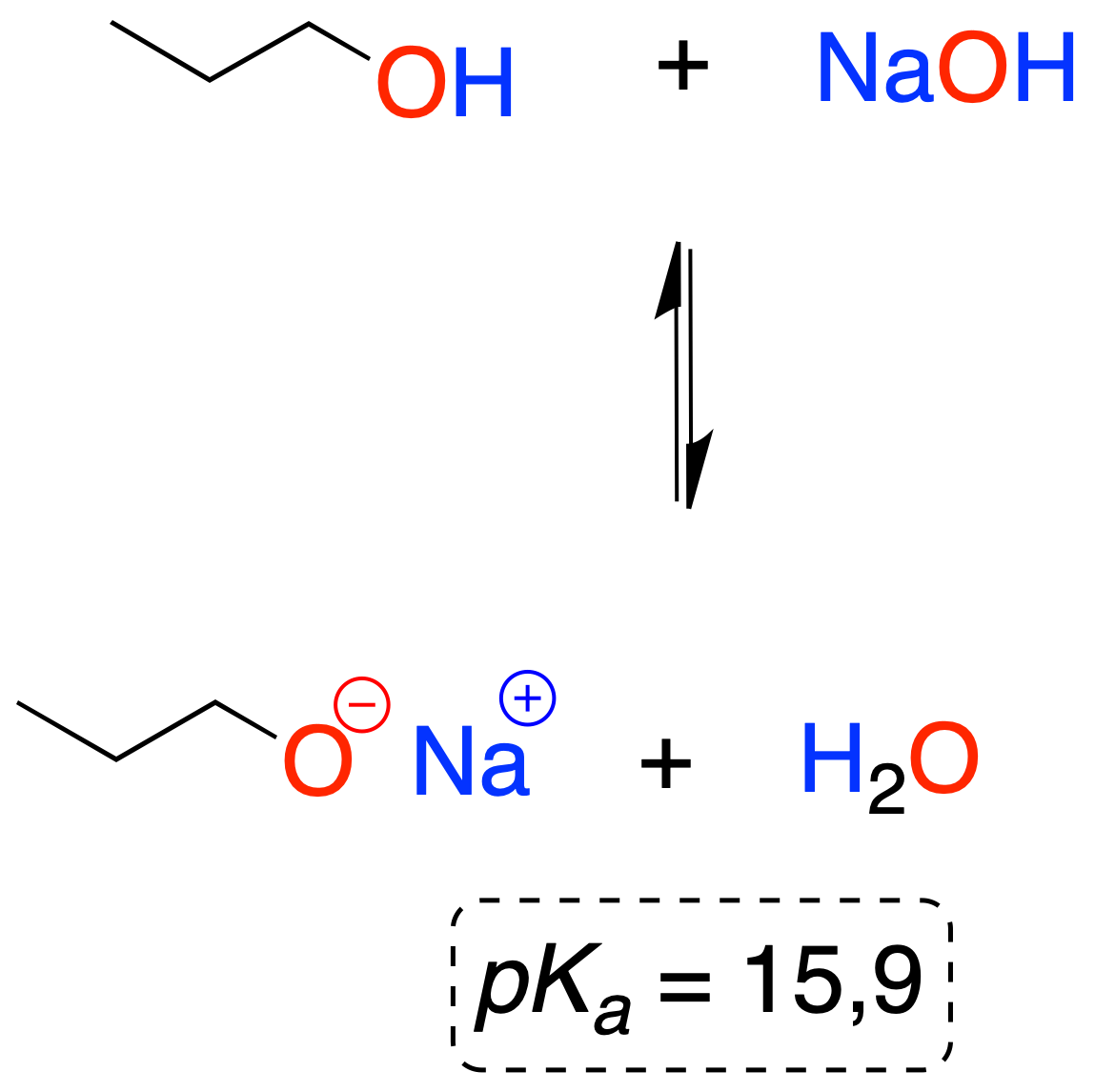
Bases such as Nao, metal hydrides, sodium amidide or butyl lithium are often used for the preparation of alkoxides.
In reactions in which alkoxide ions (R-O⊖) are involved in some way, it is sufficient that they are in a concentration lower than the stoichiometric concentration, for which sodium or potassium hydroxide (alcoholic potash) is usually added to the alcohol.
The pKa value of alcohols is in the range of 15, similar to that of water (15.7). In addition, the acidity of an alcohol depends on its structure:
CH3OH > primary (1º) > secondary (2º) > tertiary (3º)
Nomenclature
Follow the link for a summary of nomenclature of alcohols.
Reactions of alcohols
In addition to the acid-base reactions of alcohols, the main organic reactions of these functional groups are described in the section on reactions of alcohols.
Analysis of alcohols
As proof of the existence of an alcohol, one generally go to the analysis of its IR spectrum: the absence of a carbonyl band and the appearance of a band of strong intensity in the 3600 to 3400 cm-1 region.
Such a band can also be indicative of amines and phenols from which they can be distinguished by the different acidic and basic character; amines have a basic character, phenols are considerably more acidic than alcohols and dissolve in 5 % NaOH.
The following tests are used as methods for their classification and identification:
Reaction with sodium metal
Procedure: A small piece of sodium metal the size of half a pea is added over 3 ml of the alcohol and the result is observed. The test is considered positive if hydrogen release is observed.
To destroy the excess sodium, add a few milliliters of EtOH and wait for the gas release to cease before pouring into the corresponding recycling container.
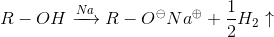
Lucas test (ZnCl2 / HCl conc)
The tertiary alcohols react readily with ZnCl2 / HCl conc. to give water-insoluble alkyl chlorides, whereas the secondary alcohols react slowly, and the primary alcohols remain practically inert. The test is not valid for aryl alcoholsand those insoluble in water.
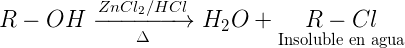
Procedure: The reagent is prepared by dissolving 16 g of anhydrous zinc chloride in 10 ml of concentrated HCl, and the mixture is cooled to avoid loss of HCl. Close the reagent flask tightly, because ZnCl2 is strongly hygroscopic and loses effectiveness. Add three or four drops of alcohol to 2 ml of Lucas’ reagent in a small test tube. Close the tube and shake vigorously for 15 s, and allow to stand at room temperature.
Depending on the result obtained, we can make the following classification:
- Tertiary alcohols, allylic alcohols and benzyl alcohols react almost immediately producing the separation of two layers (aqueous and alkyl chloride).
- The secondaries after a few minutes produce an opalescent dissolution due to the formation of insoluble chloride.
- The primaries with less than 6 carbons will dissolve and the higher ones will not dissolve and the aqueous phase will remain clear.
- If there is any doubt between secondary and tertiary, the test is repeated with concentrated HCl. The tertiaries continue to react rapidly producing insoluble alkyl chloride; while the secondaries do not react.
Oxidation test (K2Cr2O7 / H2SO4)
Primary and secondary alcohols react rapidly with chromic acid to give a greenish suspension due to the formation of Cr(III), whereas tertiary alcohols do not give this reaction.
The presence of other easily oxidizable functions such as aldehydes or phenols may interfere, since they also react with this reagent.
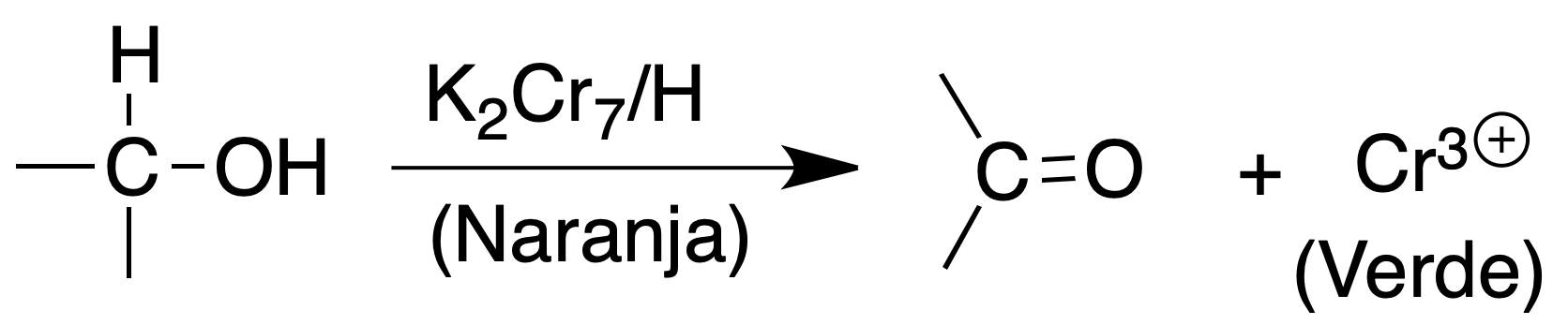
Procedure: Prepare the reagent dissolving 5 g of potassium dichromate in 50 ml of water and adding 5 ml of concentrated H2SO4. To an amount of 2 ml of the reagent a few drops of the alcohol are added, shaking the mixture.
Characterization of alcohols: obtaining esters
Numerous esters have been used as an aid in the characterization of alcohols, comprising a wide variety of phthalic acid esters, xanthates, benzoates and acetates, the last two being particularly suitable for the characterization of glycols and polyhydroxy compounds.
Acetyl chloride test (CH3COCl)
They react vigorously with acetyl chloride, forming an ester and giving off HCl which can be detected with indicator paper.
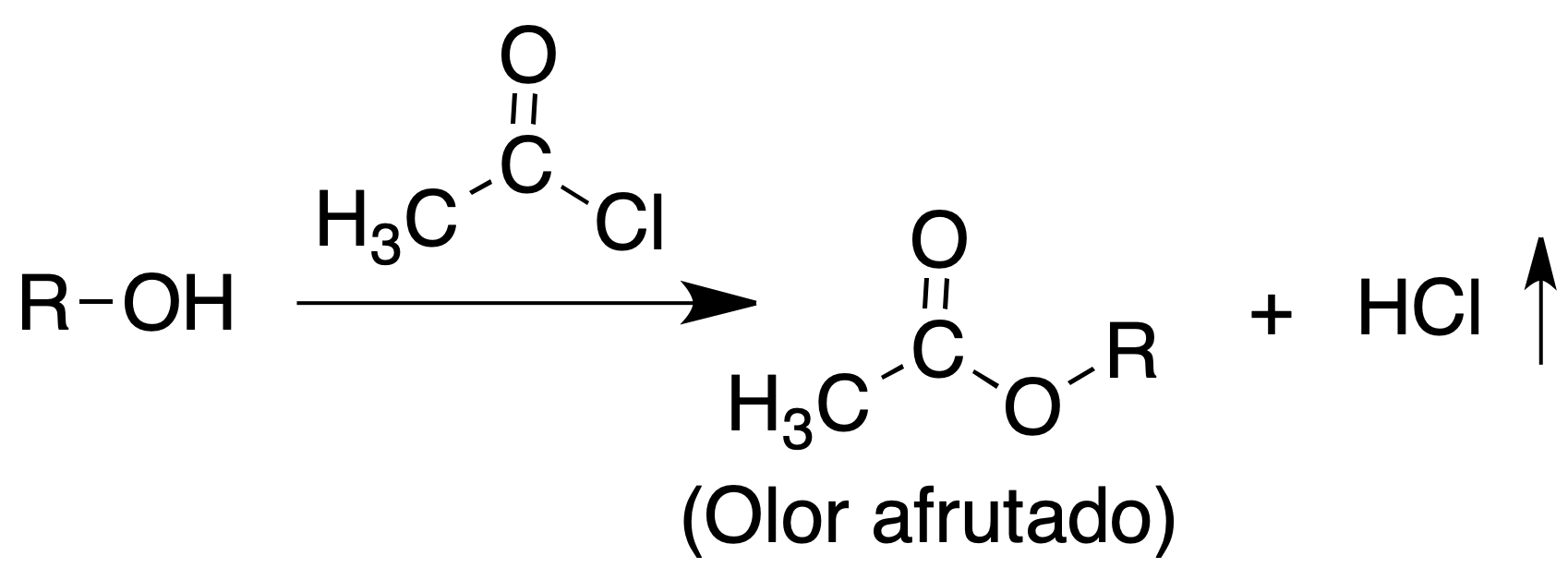
Procedure: In a dry test tube place 0.5 ml of alcohol and carefully add drop by drop 0.3 ml of acetyl chloride, observing if a reaction is produced by the heat release.
The solution is poured over 10 ml of ice-water and the odor of the sample is detected (esters of simple alcohols usually have fruity odors).
| DANGER! “Due to the hazardous nature of acetyl chloride, the test should be carried out in a fume hood wearing gloves.” |
Reaction with benzoyl chloride (Schotten-Baumann reaction)
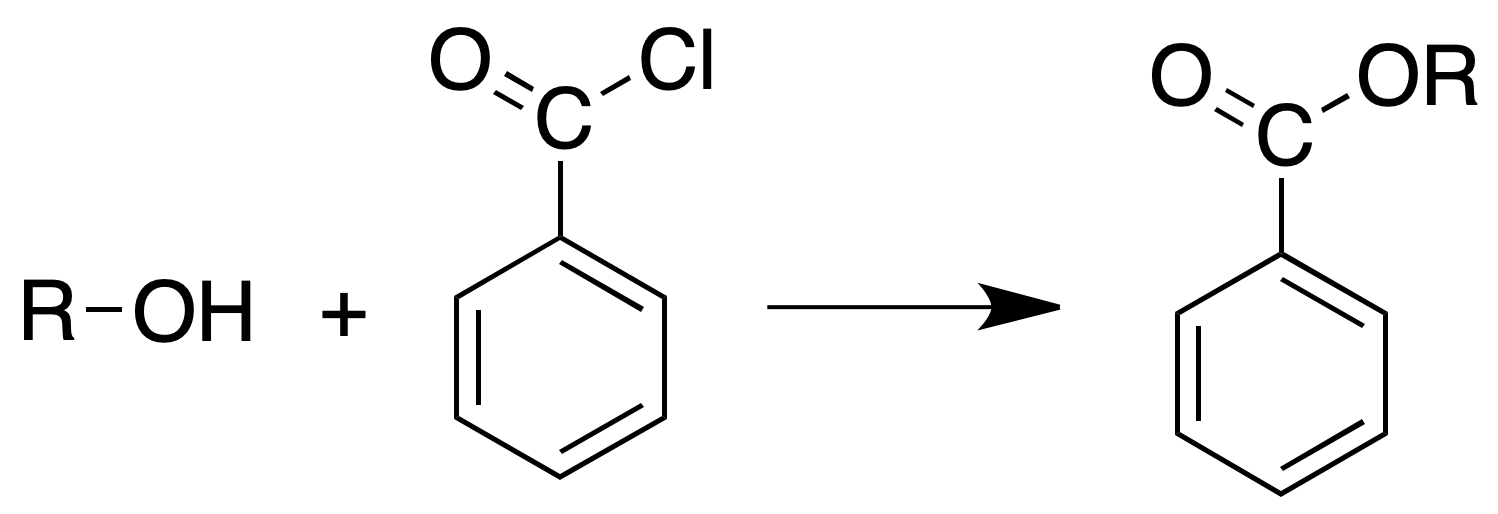
Procedure: In a test tube dissolve 2 ml of alcohol in 8 ml of 10 % NaOH. Then, in portions of 0.5 ml, a total of 2 ml of benzoyl chloride is added, shaking the test tube for 1 min after each addition.
| DANGER! “Due to the hazardous nature of benzoyl chloride, the test must be carried out in a fume hood and using gloves.” |
The reaction mixture is poured over 10 ml of cold water and the solid ester formed is collected by filtration. It can be recrystallized from EtOH/H2O.
Preparation of 3,5-dinitro and p-nitrobenzoates
If the formation of benzoates does not usually give a solid product or they are difficult to crystallize, the formation of 3,5-dinitro- and p-nitrobenzoates is used.
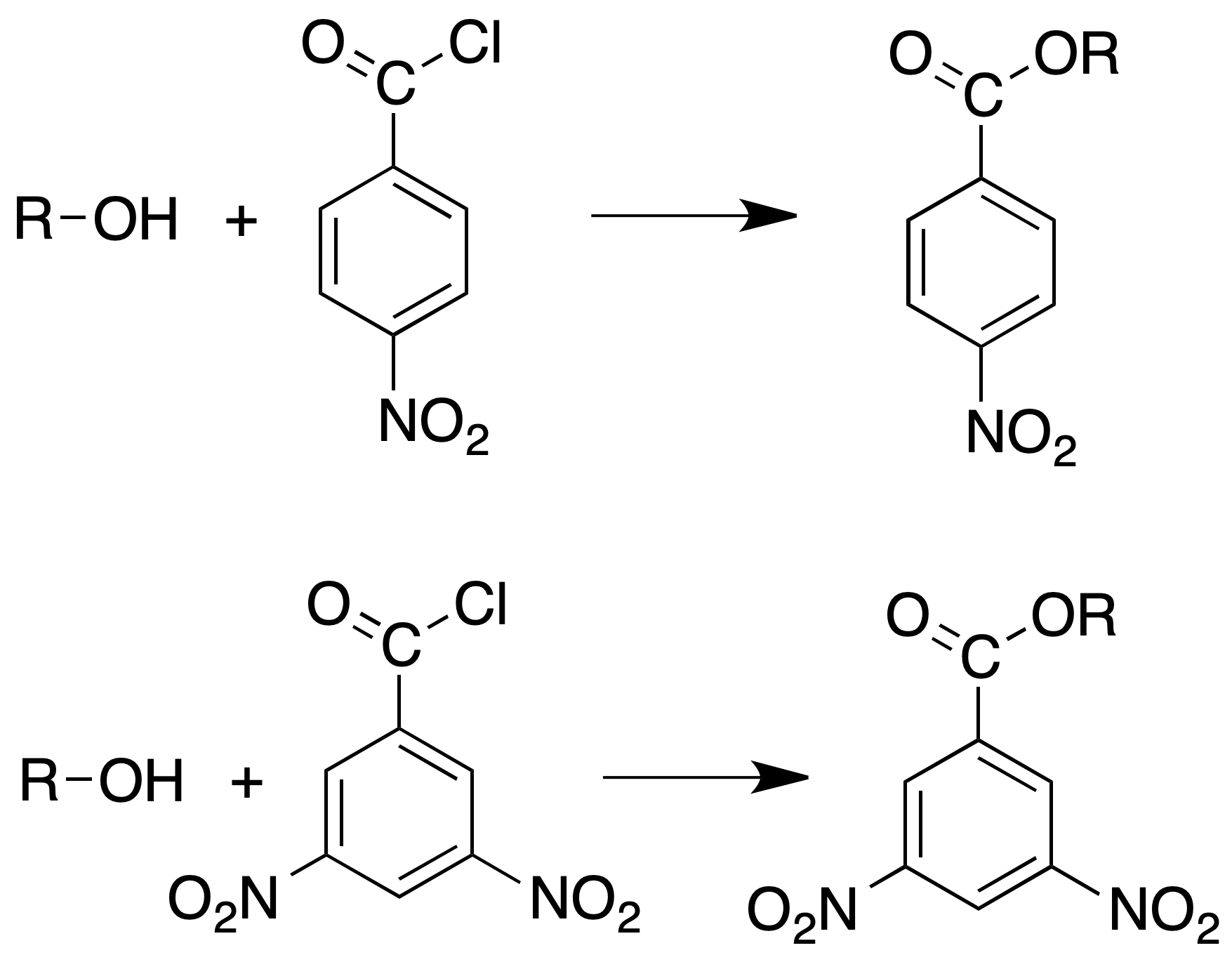
Procedure: 100 mg of alcohol, 100 mg of p-nitrobenzoyl chloride or 3,5-dinitrobenzoyl chloride, 2 ml of pyridine and a piece of porous plate are placed in a round bottom flask. Place a reflux cooler and heat for 1 h. Allow to stand overnight.
Cool the reaction mixture and add 5 ml of water and two or three drops of H2SO4. Shake vigorously and collect the crystals by filtration. Suspend the crystals in 5 ml of 2 % NaOH to remove the corresponding nitrobenzoic acid (3,5-dinitrobenzoic acid: melting point = 202 ºC; p-nitrobenzoic acid: melting point = 241 ºC). Filter, wash several times with cold water and recrystallize from EtOH or EtOH/H2O.
Characterization of alcohols: obtaining urethanes
Alcohols (must be anhydrous) react with isocyanates to produce urethanes.
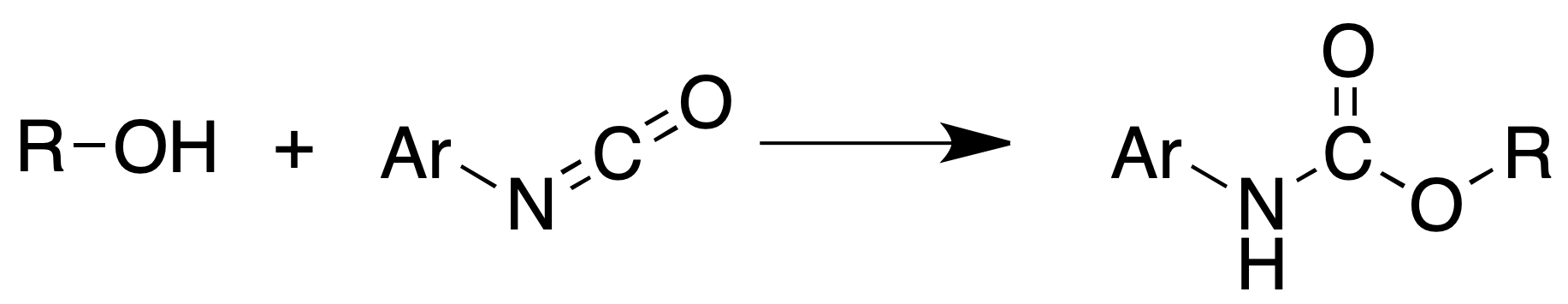
Procedure: Place 0.2 ml of alcohol and 0.2 ml of phenyl isocyanate in a dry tube. If no reaction occurs immediately, heat in a water bath at 60-70 ºC for 5 min.
Cool in an ice bath. Filter and recrystallize in petroleum ether or CCl4.
Phenols are compounds whose acidity is intermediate between that of caboxyl acids and alcohols. They can be distinguished from the latter because of their solubility in 5% NaOH solution (alcohols would not dissolve if they are not soluble in water) and they do not solubilize in 5% sodium bicarbonate solution (acids would), unless the phenol possesses highly electron-withdrawing substituents such as nitro groups.
Analysis of glycols
The physical properties of these compounds are a direct consequence of the formation of hydrogen bonds and are manifested in their relatively high boiling points, high viscosity and solubility in water.
As for their chemical properties, on the one hand they are typical of alcohols and on the other hand they are specific as a consequence of the presence of two hydroxyl groups on contiguous carbon atoms. Among these specific reactions we can highlight the following two.
Borax test
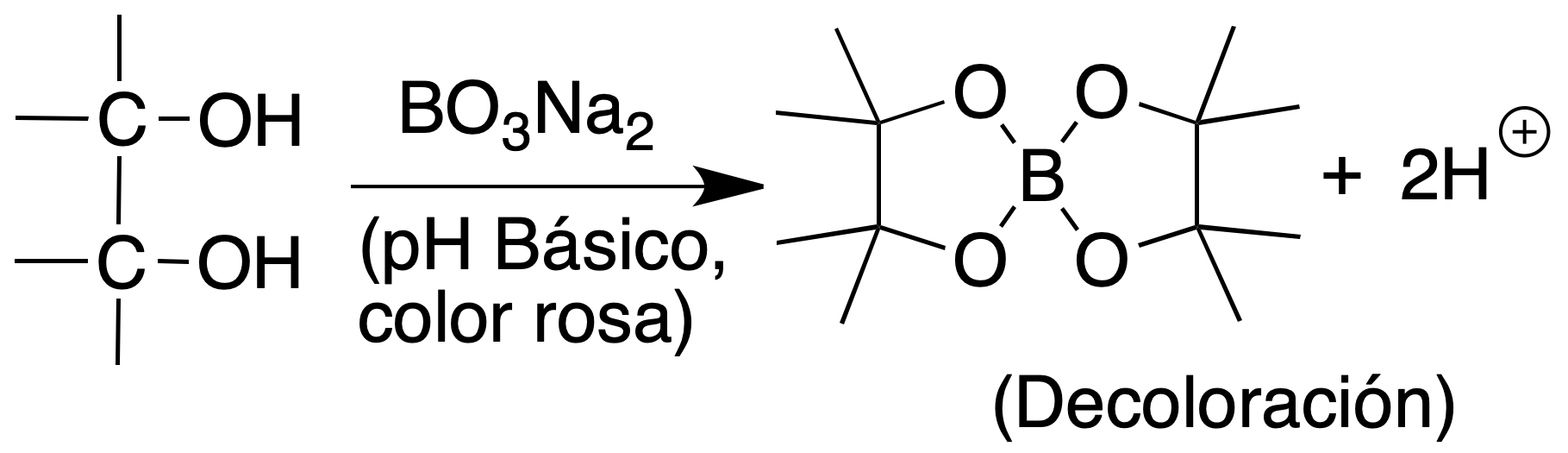
Procedure: 100 ml of 10% borax (sodium tetraborate, Na2B4O7) solution is prepared with sufficient phenolphthalein to produce a pink coloration.
Add one or two drops of the glycol to 0.5 ml of the reagent observing the disappearance of the color. The color reappears when heating and attenuates as the solution cools.
Oxidation with periodic acid (H5IO6)
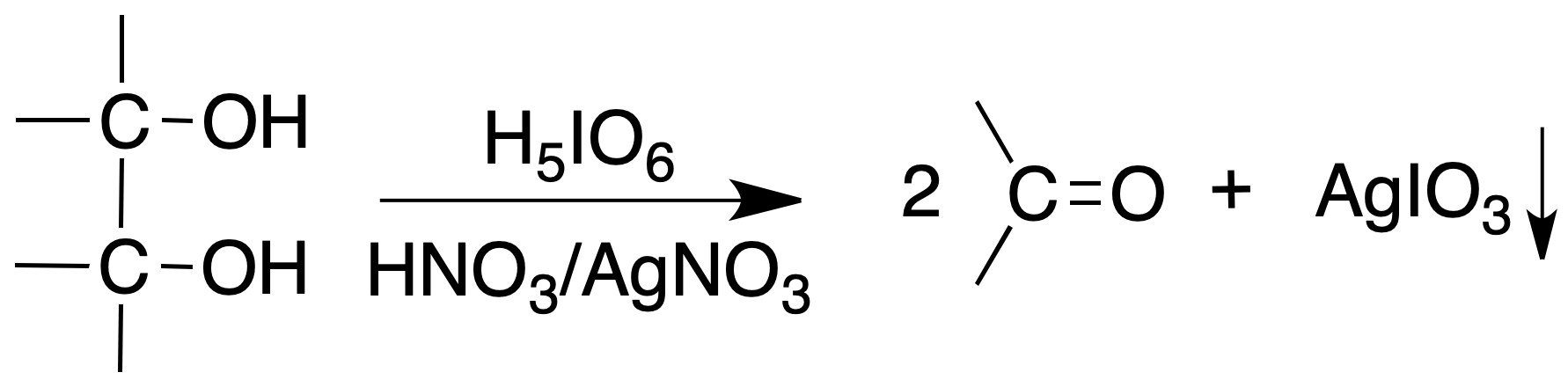
Procedure : The reagent is prepared by dissolving 0.5 g of paraperiodic acid (H5IO6) in 100 ml of deionized water. In a test tube containing 2 ml of reagent a drop of HNO3 conc. is added and shaken vigorously.
Add a drop of the compound to be tested and shake for about 15 seconds. Add one or two drops of 3 % AgNO3 solution. The instantaneous formation of a white precipitate of silver iodate means a positive result of the test.
Analysis of phenols
Ferric chloride test (FeCl3)
Most phenols give vividly colored solutions with this reagent (blue, green, violet, etc.) If the color is weak yellow, the same as that of FeCl3, the reaction is considered negative. Some phenols do not give the indicated coloration, as it happens with the hydroquinone, since it oxidizes with the reagent to quinone that does not give the coloration. The acids, with the exception of the phenolics, do not give the reaction although some give yellowish solutions or precipitates.
As a precaution it is necessary to indicate that the absence of color is not a negative test for the presence of phenol, just as a positive test should be taken with caution because other functions also give color changes: carboxylic acids, both aliphatic and aromatic, enols, oximes, hydroxamic acids and sulfinic acids.
Procedure : To 1 ml of a dilute aqueous solution of the compound several drops of FeCl3 solution at 2.5 % are added (compare the result of the coloration with a blank test). The color obtained in the test may not be permanent so it is convenient to note down the results obtained immediately after mixing.
In case the starting compound is not very soluble, it is necessary to carry out the previous operation with alcoholic solutions instead of aqueous ones.
Also, it is usually sufficient to carry out the same test using a drop of each of the reagents on a piece of filter paper and noting the color of the area where the two spots intersect.
Bromine water test
To a 1 % aqueous solution of phenol add a saturated solution of bromine water (5 ml of bromine per 100 ml of water), dropwise until the bromine color persists. A positive result will be given by the appearance of a precipitate due to the formation of bromophenol and a strongly acid reaction mixture.
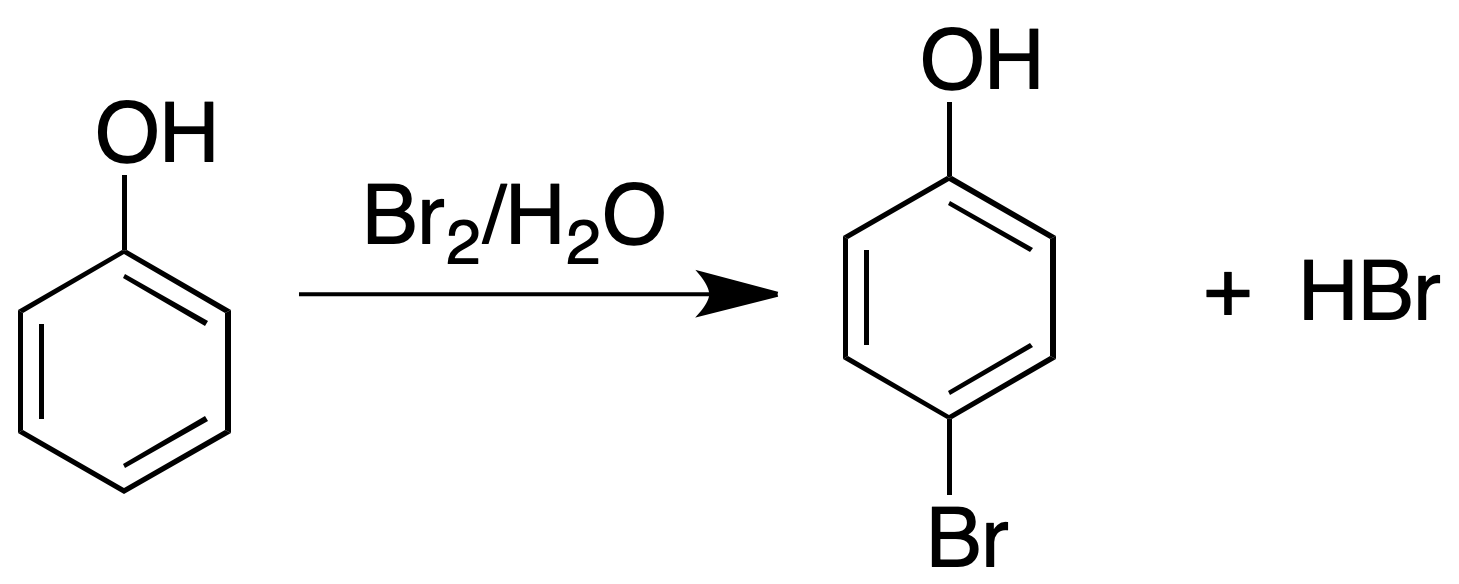
Characterization of phenols: obtaining ethers
In principle, the usual procedure is the same as for alcohols, i.e. esterification as indicated above. However, there is also the possibility of preparing ether-like compounds such as:
Aryloxyacetic acids
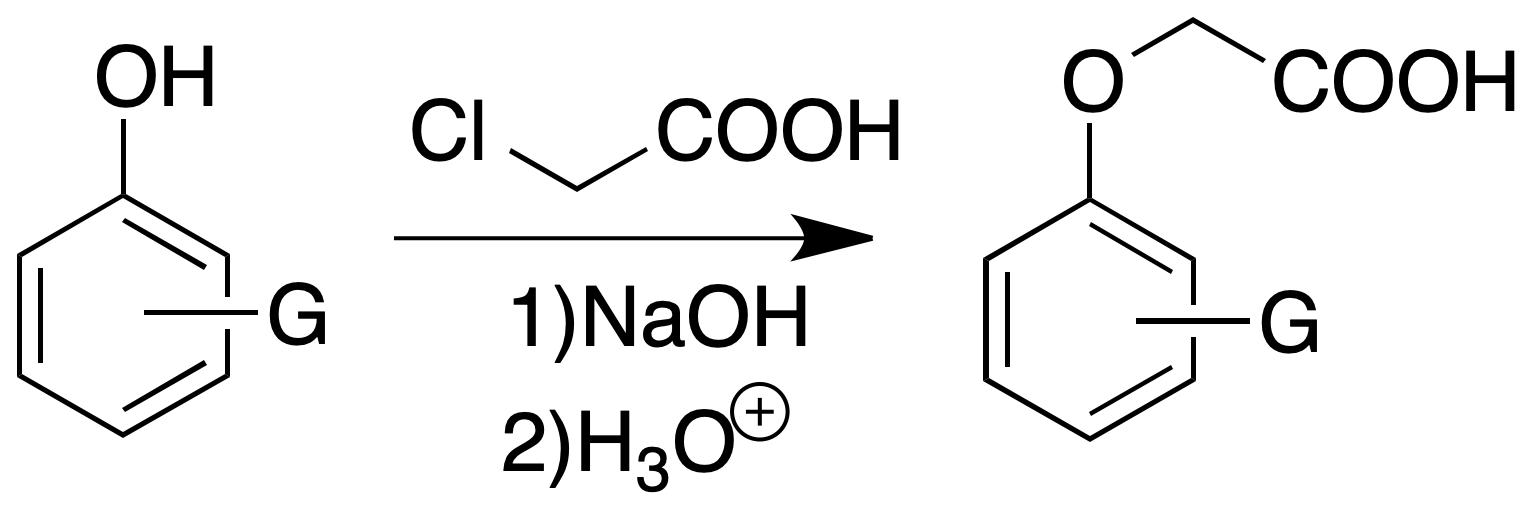
Procedure : Dissolve about 200 mg of phenol in 1 ml of 6 N sodium hydroxide (24 g of NaOH in 100 ml of water) in a small test tube. We can add a small amount of additional water to dissolve the sodium phenoxide if necessary. To this solution 0.5 ml of a 50 % aqueous solution of chloroacetic acid is added.
We put the mixture in a flask provided with reflux and we heat in a water bath of 90-100 ºC during 1 h. The solution is cooled, 2 ml of dilute HCl are added to it. The mixture is extracted several times with small portions of ethyl ether.
The ether extract is washed with water and extracted with 5% sodium carbonate solution. The extracted sodium carbonate solution is acidified with HCl to precipitate the aryloxyacetic acid. We filter and we can recrystallize from hot water.
Full Professor of Organic Chemistry at the University of Granada, with a long-standing research career in Computational Chemistry and molecular modeling and design.